Structural insights into human propionyl-CoA carboxylase (PCC) and 3-methylcrotonyl-CoA carboxylase (MCC).
Zhou, F.Y., Zhang, Y.Y., Zhu, Y., Zhou, Q., Shi, Y., Hu, Q.(2024) Elife
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
(2024) Elife
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Pyruvate carboxylase, mitochondrial | 1,178 | Homo sapiens | Mutation(s): 0 EC: 6.4.1.1 | 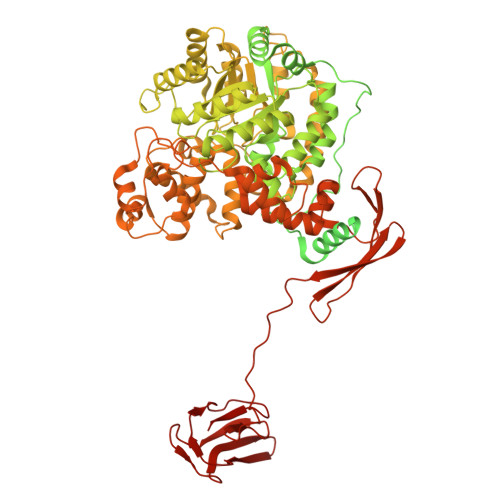 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P11498 GTEx: ENSG00000173599 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P11498 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| BTN (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | E [auth A], F [auth B], G [auth C], H [auth D] | BIOTIN C10 H16 N2 O3 S YBJHBAHKTGYVGT-ZKWXMUAHSA-N |  | ||
| Funding Organization | Location | Grant Number |
|---|---|---|
| Westlake Education Foundation | China | 101256021901 |