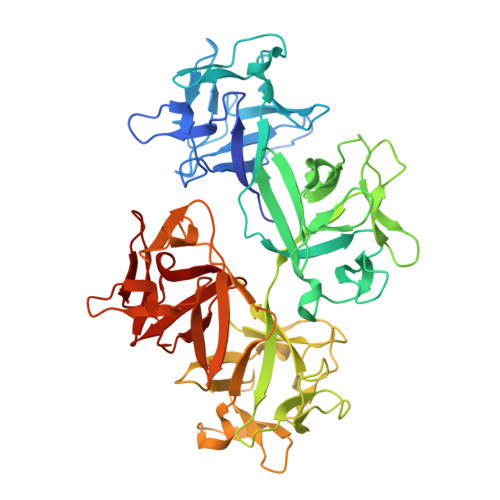
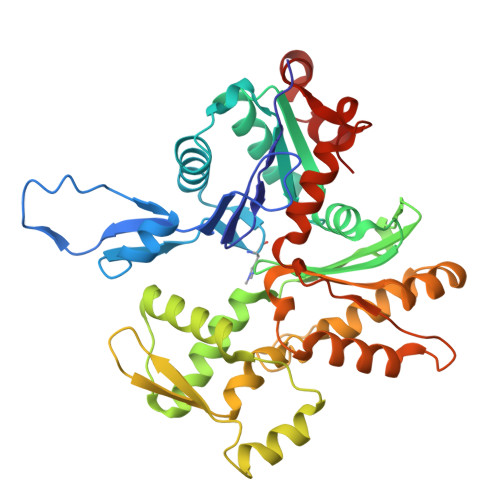
Fascin structural plasticity mediates flexible actin bundle construction.
Gong, R., Reynolds, M.J., Carney, K.R., Hamilton, K., Bidone, T.C., Alushin, G.M.(2025) Nat Struct Mol Biol 32: 940-952
- PubMed: 39833469 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41594-024-01477-2
- Primary Citation Related Structures:
8VO5, 8VO6, 8VO7, 8VO8, 8VO9, 8VOA - PubMed Abstract:
Fascin cross-links actin filaments (F-actin) into bundles that support tubular membrane protrusions including filopodia and stereocilia. Fascin dysregulation drives aberrant cell migration during metastasis, and fascin inhibitors are under development as cancer therapeutics. Here, we use cryo-EM, cryo-electron tomography coupled with custom denoising and computational modeling to probe human fascin-1's F-actin cross-linking mechanisms across spatial scales. Our fascin cross-bridge structure reveals an asymmetric F-actin binding conformation that is allosterically blocked by the inhibitor G2. Reconstructions of seven-filament hexagonal bundle elements, variability analysis and simulations show how structural plasticity enables fascin to bridge varied interfilament orientations, accommodating mismatches between F-actin's helical symmetry and bundle hexagonal packing. Tomography of many-filament bundles and modeling uncover geometric rules underlying emergent fascin binding patterns, as well as the accumulation of unfavorable cross-links that limit bundle size. Collectively, this work shows how fascin harnesses fine-tuned nanoscale structural dynamics to build and regulate micron-scale F-actin bundles.
- Laboratory of Structural Biophysics and Mechanobiology, The Rockefeller University, New York, NY, USA. rgong@rockefeller.edu.
Organizational Affiliation: