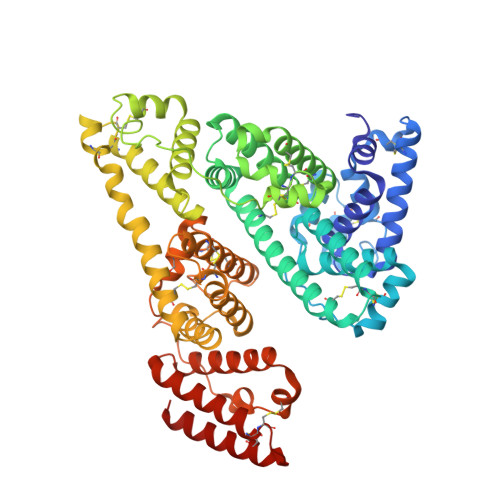
The CryoEM structure of human serum albumin in complex with ligands.
Catalano, C., Lucier, K.W., To, D., Senko, S., Tran, N.L., Farwell, A.C., Silva, S.M., Dip, P.V., Poweleit, N., Scapin, G.(2024) J Struct Biol 216: 108105-108105
- PubMed: 38852682 Search on PubMed
- DOI: https://doi.org/10.1016/j.jsb.2024.108105
- Primary Citation Related Structures:
8VAC, 8VAE, 8VAF - PubMed Abstract:
Human serum albumin (HSA) is the most prevalent plasma protein in the human body, accounting for 60 % of the total plasma protein. HSA plays a major pharmacokinetic function, serving as a facilitator in the distribution of endobiotics and xenobiotics within the organism. In this paper we report the cryoEM structures of HSA in the apo form and in complex with two ligands (salicylic acid and teniposide) at a resolution of 3.5, 3.7 and 3.4 Å, respectively. We expand upon previously published work and further demonstrate that sub-4 Å maps of ∼60 kDa proteins can be routinely obtained using a 200 kV microscope, employing standard workflows. Most importantly, these maps allowed for the identification of small molecule ligands, emphasizing the practical applicability of this methodology and providing a starting point for subsequent computational modeling and in silico optimization.
- NanoImaging Services, 4940 Carroll Canyon Road, Suite 115, San Diego, CA 92121, USA. Electronic address: ccatalano@nanoimagingservices.com.
Organizational Affiliation: