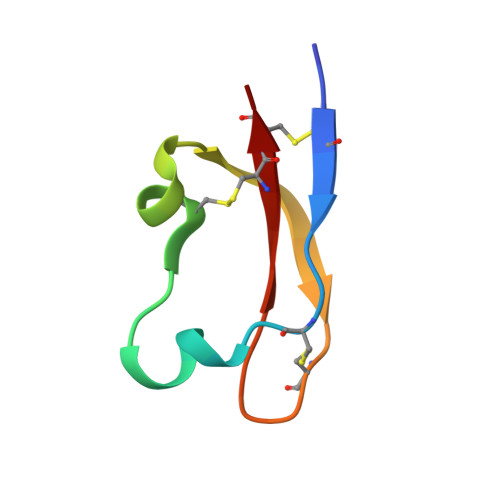
Structure and bioactivity of an insecticidal trans-defensin from assassin bug venom.
Walker, A.A., Chin, Y.K., Guo, S., Jin, J., Wilbrink, E., Goudarzi, M.H., Wirth, H., Gordon, E., Weirauch, C., King, G.F.(2024) Structure 32: 1348-1357.e4
- PubMed: 38889720
- DOI: https://doi.org/10.1016/j.str.2024.05.016
- Primary Citation of Related Structures:
8UNG - PubMed Abstract:
Disulfide-rich peptides such as defensins play diverse roles in immunity and ion channel modulation, as well as constituting the bioactive components of many animal venoms. We investigated the structure and bioactivity of U-RDTX-Pp19, a peptide previously discovered in venom of the assassin bug Pristhesancus plagipennis. Recombinant Pp19 (rPp19) was found to possess insecticidal activity when injected into Drosophila melanogaster. A bioinformatic search revealed that domains homologous to Pp19 are produced by assassin bugs and diverse other arthropods. rPp19 co-eluted with native Pp19 isolated from P. plagipennis, which we found is more abundant in hemolymph than venom. We solved the three-dimensional structure of rPp19 using 2D 1 H NMR spectroscopy, finding that it adopts a disulfide-stabilized structure highly similar to known trans-defensins, with the same cystine connectivity as human α-defensin (I-VI, II-IV, and III-V). The structure of Pp19 is unique among reported structures of arthropod peptides.
Organizational Affiliation:
Institute for Molecular Bioscience, The University of Queensland, St Lucia, QLD 4072, Australia; Centre of Excellence for Innovations in Protein and Peptide Science, St Lucia, QLD 4072, Australia. Electronic address: a.walker@imb.uq.edu.au.