Structural and Biochemical Characterization of MppQ, an L-Enduracididine Biosynthetic Enzyme from Streptomyces hygroscopicus.
Vuksanovic, N., Melkonian, T.R., Serrano, D.A., Schwabacher, A.W., Silvaggi, N.R.(2023) Biochemistry 62: 3105-3115
- PubMed: 37890134 Search on PubMed
- DOI: https://doi.org/10.1021/acs.biochem.3c00428
- Primary Citation Related Structures:
8TN2, 8TN3 - PubMed Abstract:
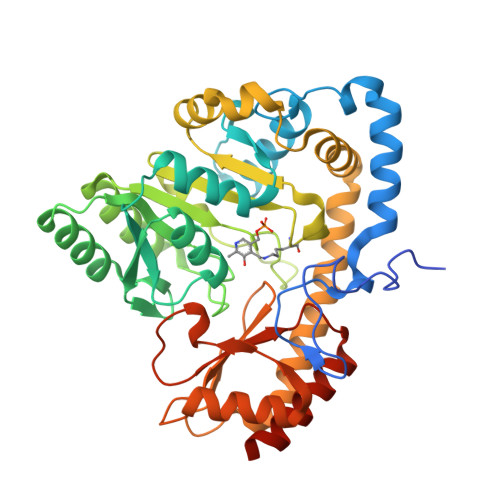
MppQ is an enzyme of unknown function from Streptomyces hygroscopicus (ShMppQ) that operates in the biosynthesis of the nonproteinogenic amino acid L-enduracididine (L-End). Since L-End is a component of several peptides showing activity against antibiotic-resistant pathogens, understanding its biosynthetic pathway could facilitate the development of chemoenzymatic routes to novel antibiotics. Herein, we report on the crystal structures of ShMppQ complexed with pyridoxal-5'-phosphate (PLP) and pyridoxamine-5'-phosphate (PMP). ShMppQ is similar to fold-type I PLP-dependent aminotransferases like aspartate aminotransferase. The tertiary structure of ShMppQ is composed of an N-terminal extension, a large domain, and a small domain. The active site is placed at the junction of the large and small domains and includes residues from both protomers of the homodimer. We also report the first functional characterization of MppQ, which we incubated with the enzymatically produced 2-ketoenduracidine and observed the conversion to L-End, establishing ShMppQ as the final enzyme in L-End biosynthesis. Additionally, we have observed that MppQ has a relatively high affinity for 2-keto-5-guanidinovaleric acid (i.e., 2-ketoarginine), a shunt product of MppP, indicating the potential role of MppQ in increasing the efficiency of L-End biosynthesis by converting 2-ketoarginine back to the starting material, l-arginine. A panel of potential amino-donor substrates was tested for the transamination activity against a saturating concentration of 2-ketoarginine in end-point assays. Most l-Arg was produced with l-ornithine as the donor substrate. Steady-state kinetic analysis of the transamination reaction with l-Orn and 2-ketoarginine shows that the kinetic constants are in line with those for the amino donor substrate of other fold-type I aminotransferases.
- Department of Chemistry and Biochemistry, University of Wisconsin-Milwaukee, 3210 North Cramer Street, Milwaukee, Wisconsin 53211, United States.
Organizational Affiliation: