Semi-invariant human type II Natural Killer T cells recognise CD1d independently of bound lipids.
Chan Yew Poa, K.T.O., Le Nours, J., Rossjohn, J., Godfrey, D.G., Almeida, C.D.S., Pellicci, D.G., Moody, B.To be published.
Experimental Data Snapshot
Starting Models: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Antigen-presenting glycoprotein CD1d | 274 | Homo sapiens | Mutation(s): 0 Gene Names: CD1D | 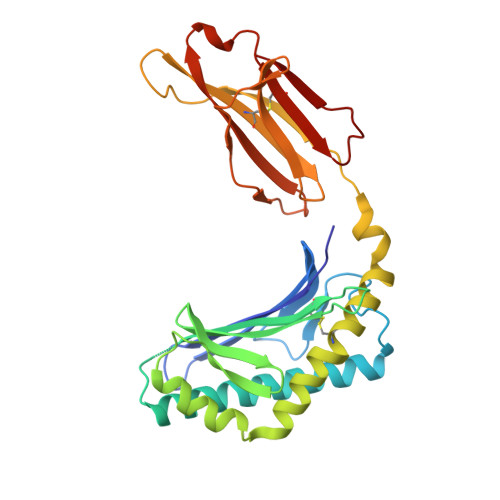 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P15813 GTEx: ENSG00000158473 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P15813 | ||||
Glycosylation | |||||
| Glycosylation Sites: 3 | Go to GlyGen: P15813-1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Beta-2-microglobulin | 100 | Homo sapiens | Mutation(s): 0 Gene Names: B2M, CDABP0092, HDCMA22P | 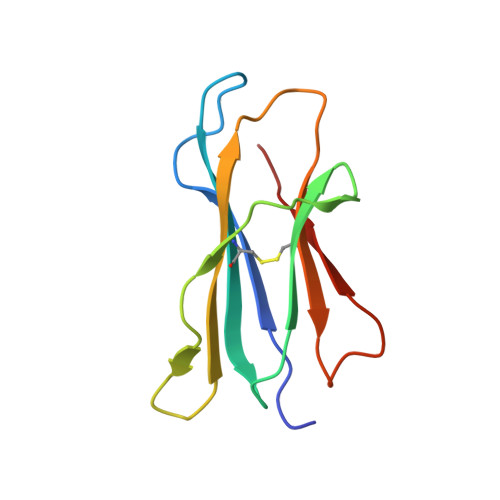 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P61769 GTEx: ENSG00000166710 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P61769 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Natural killer T cell receptor TRAV26-2 | 207 | Homo sapiens | Mutation(s): 0 | 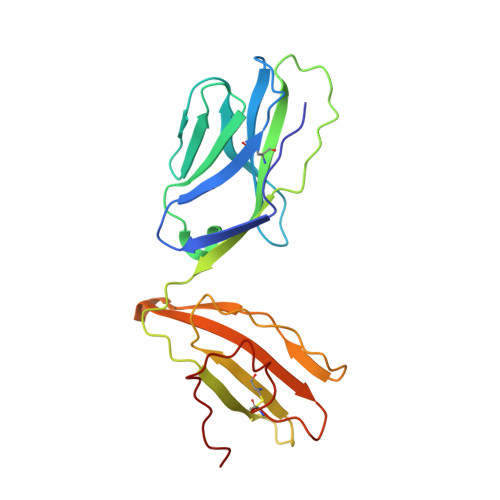 | |
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Natural killer T cell receptor TRBV19 beta chain | 243 | Homo sapiens | Mutation(s): 0 | 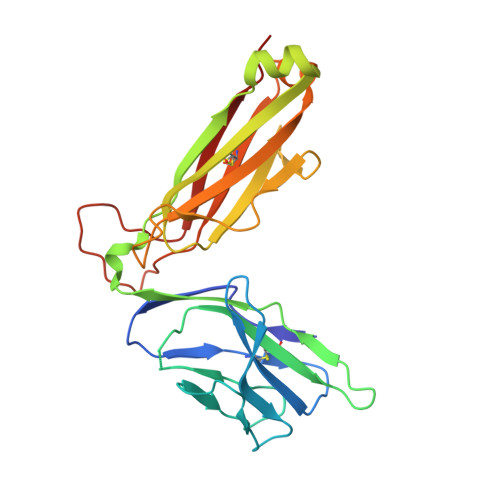 | |
Entity ID: 5 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Length | 2D Diagram | Glycosylation | D Interactions |
| 2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose | E | 2 |  | N-Glycosylation | |
Glycosylation Resources | |||||
GlyTouCan: G42666HT GlyCosmos: G42666HT GlyGen: G42666HT | |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| A1AHF (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | I [auth A] | N-[(2S)-3-oxo-1-(beta-L-talopyranosyloxy)octadecan-2-yl]docosanamide C46 H89 N O8 HKCYWBHIRZDQLD-HMGLXMNBSA-N |  | ||
| NAG Download:Ideal Coordinates CCD File | G [auth A] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
| SO4 Download:Ideal Coordinates CCD File | J [auth A], K [auth B] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| GOL Download:Ideal Coordinates CCD File | H [auth A] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 133.788 | α = 90 |
| b = 133.788 | β = 90 |
| c = 68.574 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| XDS | data reduction |
| SCALA | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Health and Medical Research Council (NHMRC, Australia) | Australia | -- |