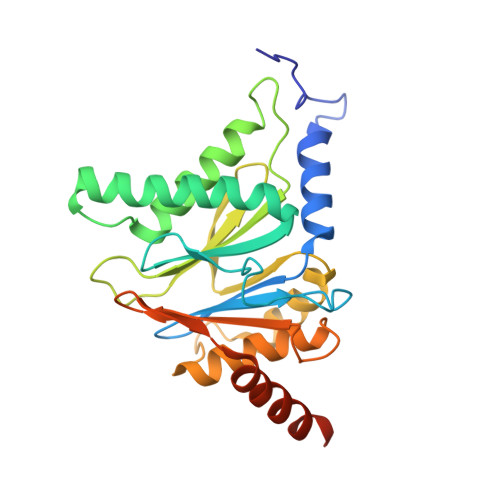
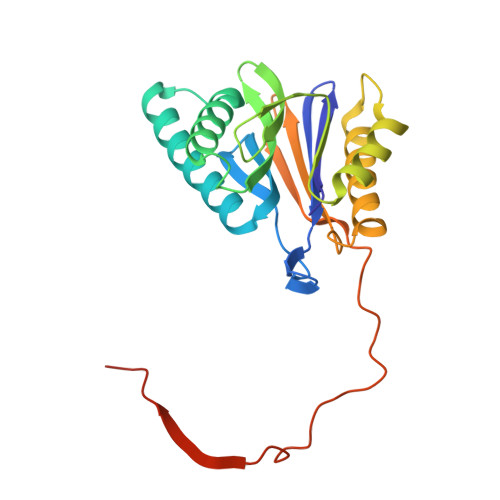
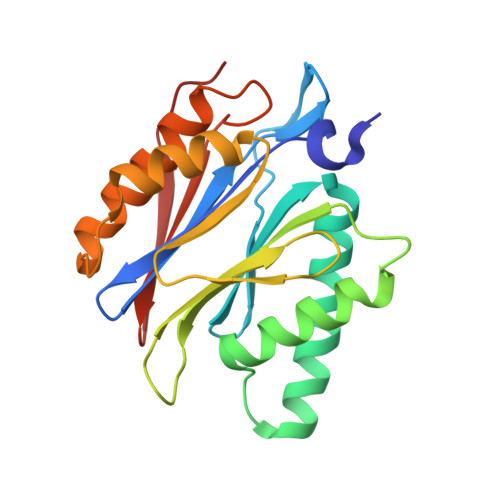
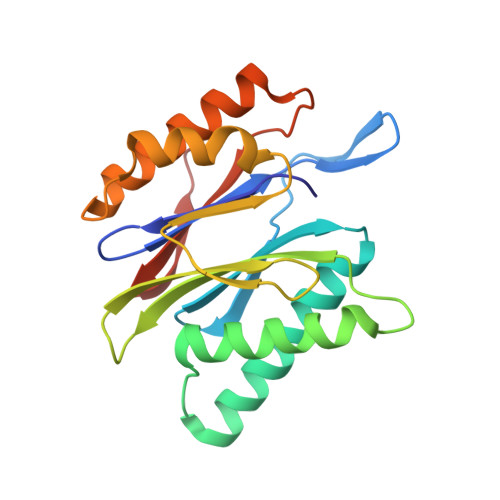
Macrocyclic Oxindole Peptide Epoxyketones-A Comparative Study of Macrocyclic Inhibitors of the 20S Proteasome.
Gotz, M.G., Godwin, K., Price, R., Dorn, R., Merrill-Steskal, G., Klemmer, W., Hansen, H., Produturi, G., Rocha, M., Palmer, M., Molacek, L., Strater, Z., Groll, M.(2024) ACS Med Chem Lett 15: 533-539
- PubMed: 38628795 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1021/acsmedchemlett.4c00017
- Primary Citation Related Structures:
8RHJ, 8RHK, 8RHL - PubMed Abstract:
Peptide macrocycles have recently gained attention as protease inhibitors due to their metabolic stability and specificity. However, the development of peptide macrocycles with improved binding potency has so far been challenging. Here we present macrocyclic peptides derived from the clinically applied proteasome inhibitor carfilzomib with an oxindole group that mimics the natural product TMC-95A. Fluorescence kinetic activity assays reveal a high potency of the oxindole group (IC 50 = 0.19 μM) compared with agents lacking this motif. X-ray structures of the ligands with the β5-subunit of the yeast 20S proteasome illustrate that the installed macrocycle forces strong hydrogen bonding of the oxindole group with β5-Gly23NH. Thus, the binding of our designed oxindole epoxyketones is entropically and enthalpically favored in contrast to more flexible proteasome inhibitors such as carfilzomib.
- Department of Chemistry, Whitman College, Walla Walla, Washington 99362, United States.
Organizational Affiliation: