Mechanism of Two-Component Mono-Oxygenases Involved in Anthracycline 1-Hydroxylation
Wandi, B.N., Dinis, P., Siitonen, V., Schneider, G., Schnell, R., Metsa-Ketele, M.(2024) ACS Catal
Experimental Data Snapshot
(2024) ACS Catal
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| SnoL | 148 | Streptomyces nogalater | Mutation(s): 0 Gene Names: snoL | 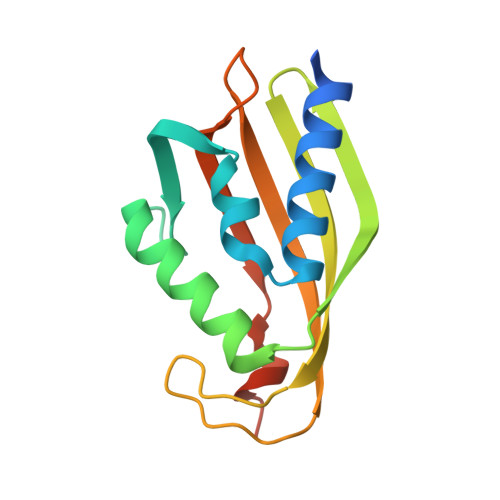 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9RN64 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| XN0 (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | C [auth A] | 3',4'-demethoxy-nogalose-nogalamycinone C29 H32 O12 XDICDWFRDRDUMQ-GUXZDVRLSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 52.628 | α = 90 |
| b = 62.281 | β = 90 |
| c = 89.814 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| Aimless | data scaling |
| XDS | data reduction |
| MOLREP | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Swedish Research Council | Sweden | 2018-03999 |