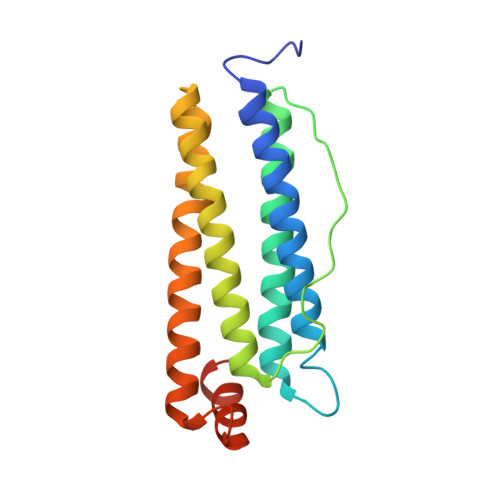
Structural basis for the intracellular regulation of ferritin degradation.
Hoelzgen, F., Nguyen, T.T.P., Klukin, E., Boumaiza, M., Srivastava, A.K., Kim, E.Y., Zalk, R., Shahar, A., Cohen-Schwartz, S., Meyron-Holtz, E.G., Bou-Abdallah, F., Mancias, J.D., Frank, G.A.(2024) Nat Commun 15: 3802-3802
- PubMed: 38714719 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-024-48151-1
- Primary Citation Related Structures:
8QU9 - PubMed Abstract:
The interaction between nuclear receptor coactivator 4 (NCOA4) and the iron storage protein ferritin is a crucial component of cellular iron homeostasis. The binding of NCOA4 to the FTH1 subunits of ferritin initiates ferritinophagy-a ferritin-specific autophagic pathway leading to the release of the iron stored inside ferritin. The dysregulation of NCOA4 is associated with several diseases, including neurodegenerative disorders and cancer, highlighting the NCOA4-ferritin interface as a prime target for drug development. Here, we present the cryo-EM structure of the NCOA4-FTH1 interface, resolving 16 amino acids of NCOA4 that are crucial for the interaction. The characterization of mutants, designed to modulate the NCOA4-FTH1 interaction, is used to validate the significance of the different features of the binding site. Our results explain the role of the large solvent-exposed hydrophobic patch found on the surface of FTH1 and pave the way for the rational development of ferritinophagy modulators.
- The Kreitman School of Advanced Graduate Studies, Marcus Family Campus, Ben-Gurion University of the Negev, Beer-Sheva, Israel.
Organizational Affiliation: