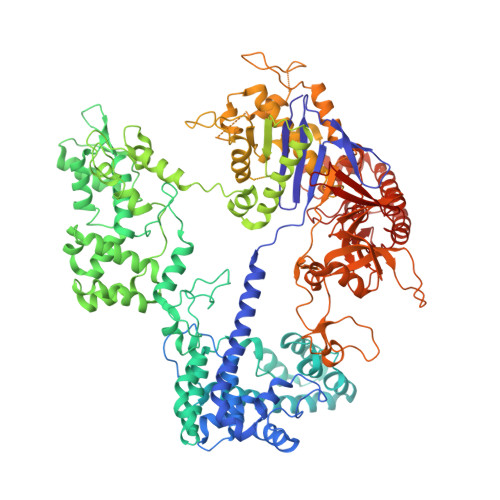
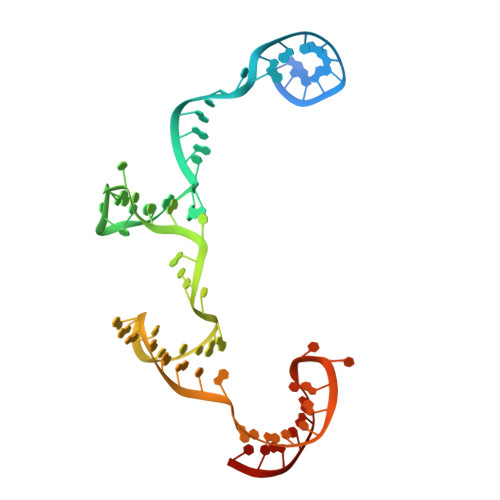
Structural insights into Cas9-mediated prespacer selection in CRISPR-Cas adaptation.
Gaizauskaite, U., Tamulaitiene, G., Silanskas, A., Gasiunas, G., Siksnys, V., Sasnauskas, G.(2026) Mol Cell
- PubMed: 41702403 Search on PubMed
- DOI: https://doi.org/10.1016/j.molcel.2026.01.022
- Primary Citation Related Structures:
8PJ9, 9H1H, 9H1V, 9H21, 9H2G, 9H2M, 9H6T, 9H72, 9HP8, 9HP9, 9Q85 - PubMed Abstract:
During CRISPR-Cas adaptation, prokaryotic cells become immunized by the insertion of foreign DNA fragments, termed spacers, into the host genome to serve as templates for RNA-guided immunity. Spacer acquisition relies on the Cas1-Cas2 integrase and accessory proteins, which select DNA sequences flanked by the protospacer adjacent motif (PAM) and insert them into the CRISPR array. It has been shown that in type II-A systems, selection of PAM-proximal prespacers is mediated by the effector nuclease Cas9, which forms a "supercomplex" with the Cas1-Cas2 integrase and the Csn2 protein. Here, we present cryo-electron microscopy structures of the Streptococcus thermophilus type II-A prespacer selection supercomplex in the DNA-scanning and two distinct PAM-bound configurations, providing insights into the mechanism of Cas9-mediated prespacer selection in type II-A CRISPR-Cas systems. Repurposing Cas9 by the CRISPR adaptation machinery for prespacer selection, as characterized here, demonstrates Cas9 plasticity and expands our knowledge of Cas9 biology.
- Institute of Biotechnology, Life Sciences Center, Vilnius University, Saulėtekio av. 7, 10257 Vilnius, Lithuania.
Organizational Affiliation: