Crystal structure of glutathione S-transferase Tau7 from Salix lindleyana in complex with GSH
Xu, N.To be published.
Experimental Data Snapshot
Starting Model: in silico
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| glutathione transferase | 222 | Salix lindleyana | Mutation(s): 0 Gene Names: DKX38_015263 EC: 2.5.1.18 | 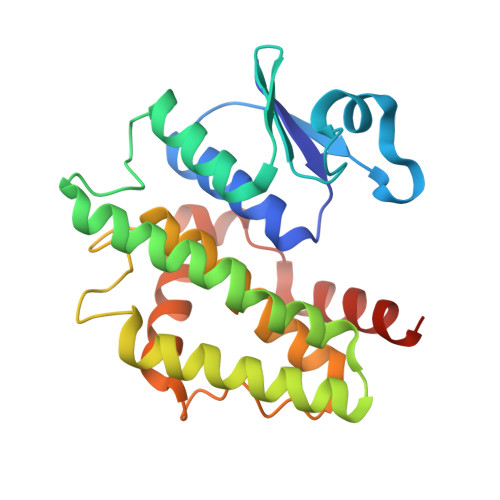 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A5N5L538 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| GSH (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | B [auth A] | Glutathione C10 H17 N3 O6 S RWSXRVCMGQZWBV-WDSKDSINSA-N |  | ||
| Entity ID: 2 | |||||
|---|---|---|---|---|---|
| ID | Chains | Name | Type/Class | 2D Diagram | 3D Interactions |
| PRD_002593 (GSH) Query on PRD_002593 | B [auth A] | Glutathione | Peptide-like / Oxidation-reduction |  | |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 60.727 | α = 90 |
| b = 87.931 | β = 90 |
| c = 113.973 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| xia2 | data reduction |
| xia2 | data scaling |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Natural Science Foundation of China (NSFC) | China | -- |