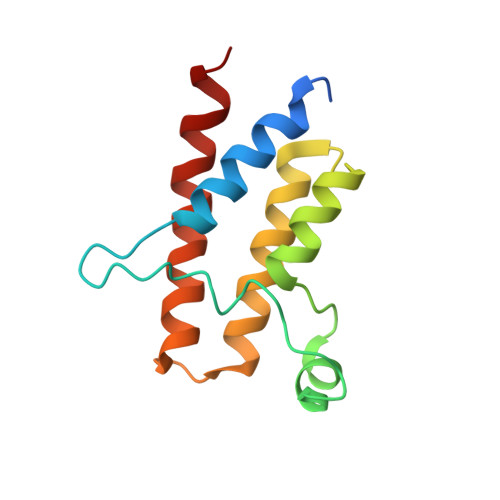
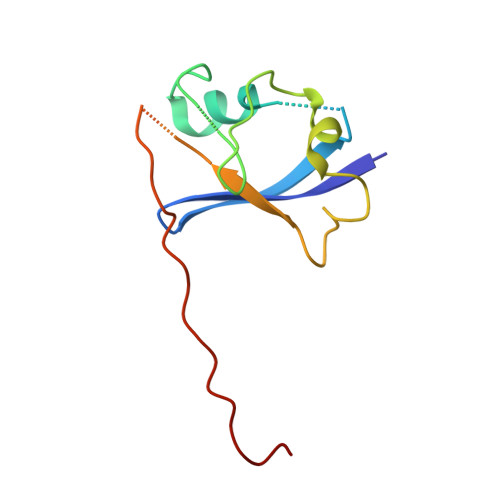
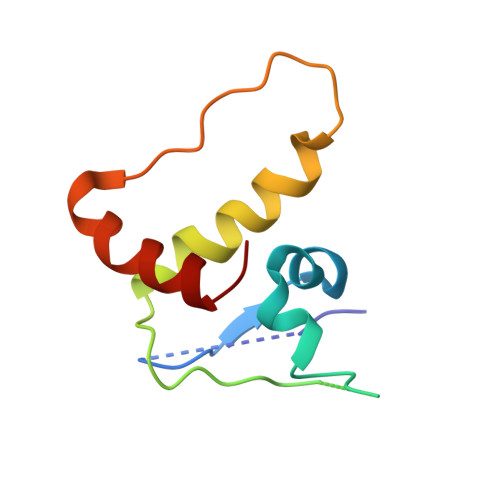
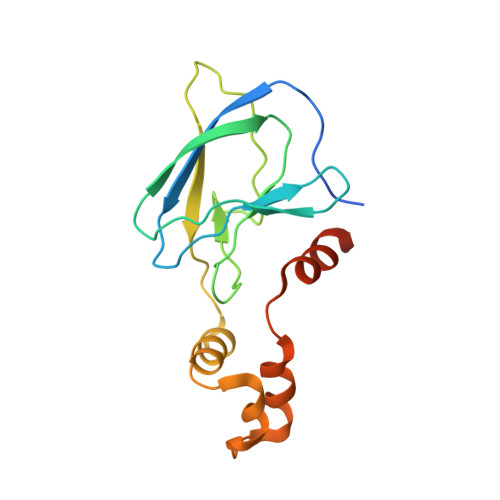
Affinity and cooperativity modulate ternary complex formation to drive targeted protein degradation.
Wurz, R.P., Rui, H., Dellamaggiore, K., Ghimire-Rijal, S., Choi, K., Smither, K., Amegadzie, A., Chen, N., Li, X., Banerjee, A., Chen, Q., Mohl, D., Vaish, A.(2023) Nat Commun 14: 4177-4177
- PubMed: 37443112 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-023-39904-5
- Primary Citation Related Structures:
8G1P, 8G1Q - PubMed Abstract:
Targeted protein degradation via "hijacking" of the ubiquitin-proteasome system using proteolysis targeting chimeras (PROTACs) has evolved into a novel therapeutic modality. The design of PROTACs is challenging; multiple steps involved in PROTAC-induced degradation make it difficult to establish coherent structure-activity relationships. Herein, we characterize PROTAC-mediated ternary complex formation and degradation by employing von Hippel-Lindau protein (VHL) recruiting PROTACs for two different target proteins, SMARCA2 and BRD4. Ternary-complex attributes and degradation activity parameters are evaluated by varying components of the PROTAC's architecture. Ternary complex binding affinity and cooperativity correlates well with degradation potency and initial rates of degradation. Additionally, we develop a ternary-complex structure modeling workflow to calculate the total buried surface area at the interface, which is in agreement with the measured ternary complex binding affinity. Our findings establish a predictive framework to guide the design of potent degraders.
- Amgen Research, Amgen Inc., Thousand Oaks, CA, USA.
Organizational Affiliation: