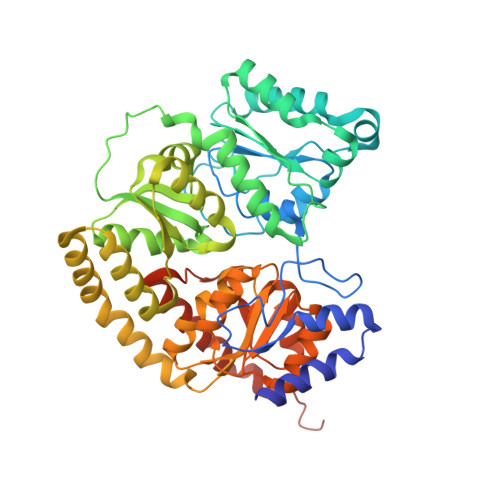
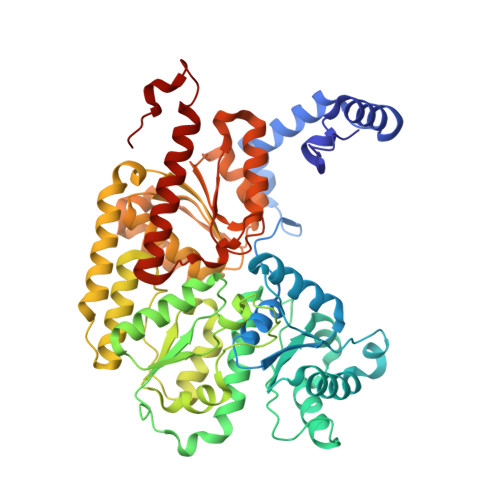
Role of Serine Coordination in the Structural and Functional Protection of the Nitrogenase P-Cluster.
Rutledge, H.L., Field, M.J., Rittle, J., Green, M.T., Tezcan, F.A.(2022) J Am Chem Soc 144: 22101-22112
- PubMed: 36445204 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1021/jacs.2c09480
- Primary Citation Related Structures:
8E3T, 8E3U, 8E3V - PubMed Abstract:
Nitrogenase catalyzes the multielectron reduction of dinitrogen to ammonia. Electron transfer in the catalytic protein (MoFeP) proceeds through a unique [8Fe-7S] cluster (P-cluster) to the active site (FeMoco). In the reduced, all-ferrous (P N ) state, the P-cluster is coordinated by six cysteine residues. Upon two-electron oxidation to the P 2+ state, the P-cluster undergoes conformational changes in which a highly conserved oxygen-based residue (a Ser or a Tyr) and a backbone amide additionally ligate the cluster. Previous studies of Azotobacter vinelandii ( Av ) MoFeP revealed that when the oxygen-based residue, βSer188, was mutated to a noncoordinating residue, Ala, the P-cluster became redox-labile and reversibly lost two of its eight Fe centers. Surprisingly, the Av strain with a MoFeP variant that lacked the serine ligand ( Av βSer188Ala MoFeP) displayed the same diazotrophic growth and in vitro enzyme turnover rates as wild-type Av MoFeP, calling into question the necessity of this conserved ligand for nitrogenase function. Based on these observations, we hypothesized that βSer188 plays a role in protecting the P-cluster under nonideal conditions. Here, we investigated the protective role of βSer188 both in vivo and in vitro by characterizing the ability of Av βSer188Ala cells to grow under suboptimal conditions (high oxidative stress or Fe limitation) and by determining the tendency of βSer188Ala MoFeP to be mismetallated in vitro . Our results demonstrate that βSer188 (1) increases Av cell survival upon exposure to oxidative stress in the form of hydrogen peroxide, (2) is necessary for efficient Av diazotrophic growth under Fe-limiting conditions, and (3) may protect the P-cluster from metal exchange in vitro . Taken together, our findings suggest a structural adaptation of nitrogenase to protect the P-cluster via Ser ligation, which is a previously unidentified functional role of the Ser residue in redox proteins and adds to the expanding functional roles of non-Cys ligands to FeS clusters.
- Department of Chemistry and Biochemistry, University of California, San Diego, 9500 Gilman Drive, La Jolla, California 92093-0340, United States.
Organizational Affiliation: