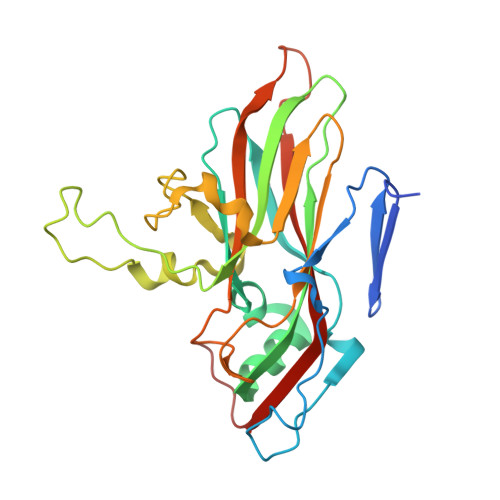
A tradeoff between enterovirus A71 particle stability and cell entry.
Catching, A., Te Yeh, M., Bianco, S., Capponi, S., Andino, R.(2023) Nat Commun 14: 7450-7450
- PubMed: 37978288 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-023-43029-0
- Primary Citation Related Structures:
8E2X, 8E2Y, 8E31, 8E38, 8E39, 8E3A, 8E3B, 8E3C - PubMed Abstract:
A central role of viral capsids is to protect the viral genome from the harsh extracellular environment while facilitating initiation of infection when the virus encounters a target cell. Viruses are thought to have evolved an optimal equilibrium between particle stability and efficiency of cell entry. In this study, we genetically perturb this equilibrium in a non-enveloped virus, enterovirus A71 to determine its structural basis. We isolate a single-point mutation variant with increased particle thermotolerance and decreased efficiency of cell entry. Using cryo-electron microscopy and molecular dynamics simulations, we determine that the thermostable native particles have acquired an expanded conformation that results in a significant increase in protein dynamics. Examining the intermediate states of the thermostable variant reveals a potential pathway for uncoating. We propose a sequential release of the lipid pocket factor, followed by internal VP4 and ultimately the viral RNA.
- Department of Microbiology and Immunology, University of California in San Francisco, San Francisco, CA, 94158, USA.
Organizational Affiliation: