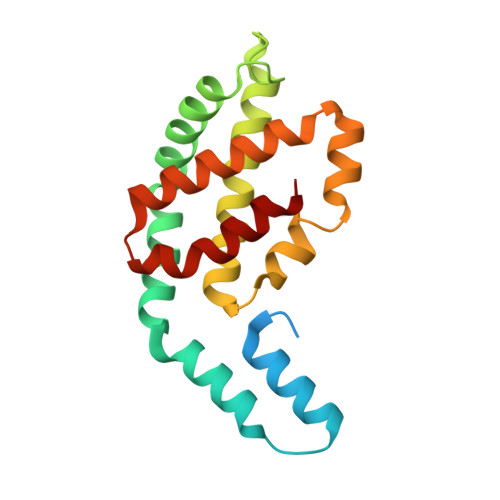
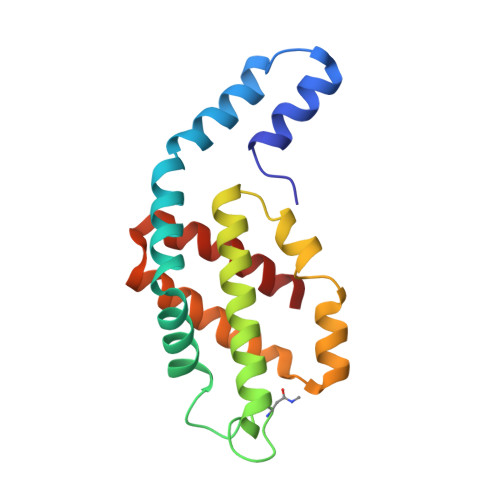
Helical allophycocyanin nanotubes absorb far-red light in a thermophilic cyanobacterium.
Gisriel, C.J., Elias, E., Shen, G., Soulier, N.T., Flesher, D.A., Gunner, M.R., Brudvig, G.W., Croce, R., Bryant, D.A.(2023) Sci Adv 9: eadg0251-eadg0251
- PubMed: 36961897 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1126/sciadv.adg0251
- Primary Citation Related Structures:
8DDY - PubMed Abstract:
To compete in certain low-light environments, some cyanobacteria express a paralog of the light-harvesting phycobiliprotein, allophycocyanin (AP), that strongly absorbs far-red light (FRL). Using cryo-electron microscopy and time-resolved absorption spectroscopy, we reveal the structure-function relationship of this FRL-absorbing AP complex (FRL-AP) that is expressed during acclimation to low light and that likely associates with chlorophyll a-containing photosystem I. FRL-AP assembles as helical nanotubes rather than typical toroids due to alterations of the domain geometry within each subunit. Spectroscopic characterization suggests that FRL-AP nanotubes are somewhat inefficient antenna; however, the enhanced ability to harvest FRL when visible light is severely attenuated represents a beneficial trade-off. The results expand the known diversity of light-harvesting proteins in nature and exemplify how biological plasticity is achieved by balancing resource accessibility with efficiency.
- Department of Chemistry, Yale University, New Haven, CT 06520, USA.
Organizational Affiliation: