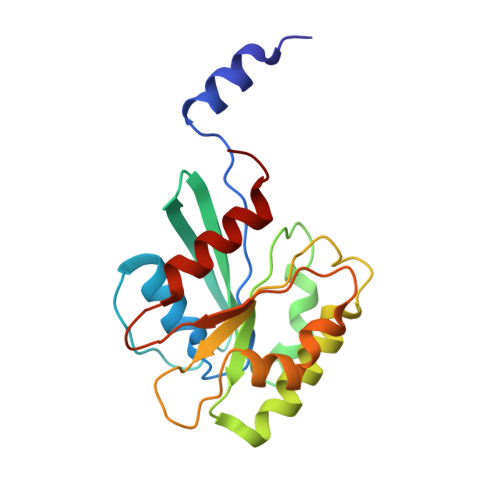
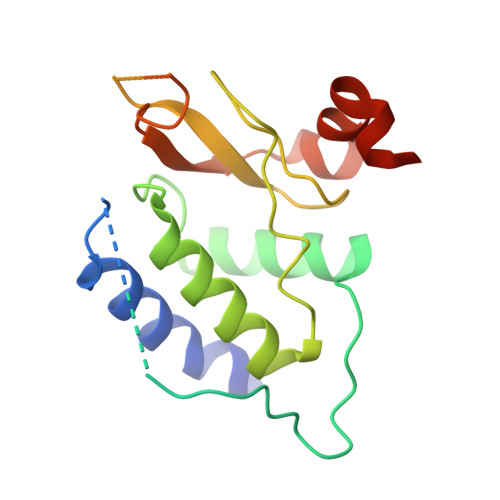
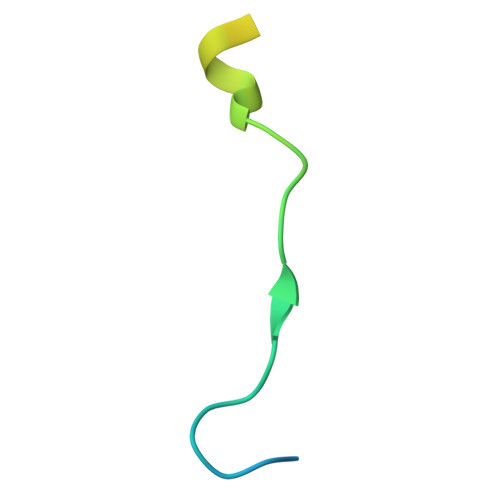
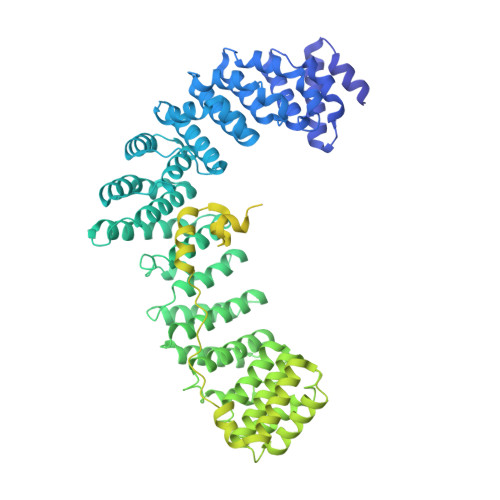
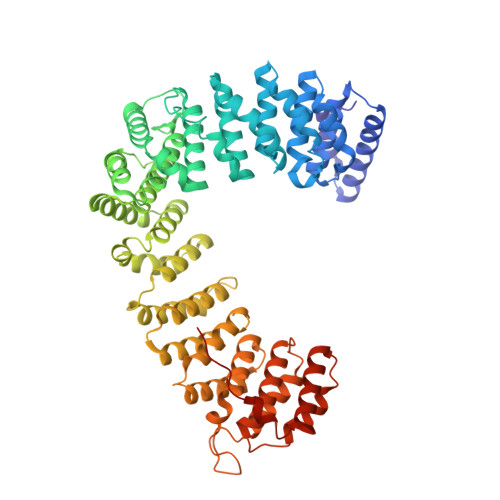
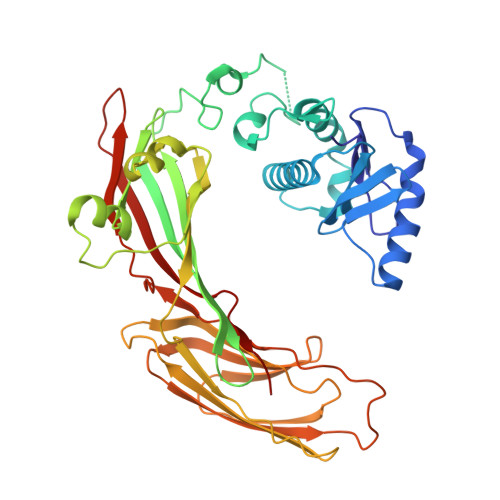
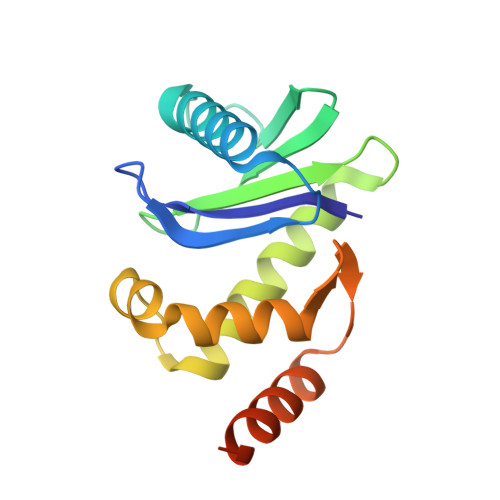
Self-assembly and structure of a clathrin-independent AP-1:Arf1 tubular membrane coat.
Hooy, R.M., Iwamoto, Y., Tudorica, D.A., Ren, X., Hurley, J.H.(2022) Sci Adv 8: eadd3914-eadd3914
- PubMed: 36269825 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1126/sciadv.add3914
- Primary Citation Related Structures:
7UX3, 8D4C, 8D4D, 8D4E, 8D4F, 8D4G, 8D9R, 8D9S, 8D9T, 8D9U, 8D9V, 8D9W - PubMed Abstract:
The adaptor protein (AP) complexes not only form the inner layer of clathrin coats but also have clathrin-independent roles in membrane traffic whose mechanisms are unknown. HIV-1 Nef hijacks AP-1 to sequester major histocompatibility complex class I (MHC-I), evading immune detection. We found that AP-1:Arf1:Nef:MHC-I forms a coat on tubulated membranes without clathrin and determined its structure. The coat assembles via Arf1 dimer interfaces. AP-1-positive tubules are enriched in cells upon clathrin knockdown. Nef localizes preferentially to AP-1 tubules in cells, explaining how Nef sequesters MHC-I. Coat contact residues are conserved across Arf isoforms and the Arf-dependent AP complexes AP-1, AP-3, and AP-4. Thus, AP complexes can self-assemble with Arf1 into tubular coats without clathrin or other scaffolding factors. The AP-1:Arf1 coat defines the structural basis of a broader class of tubulovesicular membrane coats as an intermediate in clathrin vesicle formation from internal membranes and as an MHC-I sequestration mechanism in HIV-1 infection.
- Department of Molecular and Cell Biology, University of California, Berkeley, Berkeley, CA 94720, USA.
Organizational Affiliation: