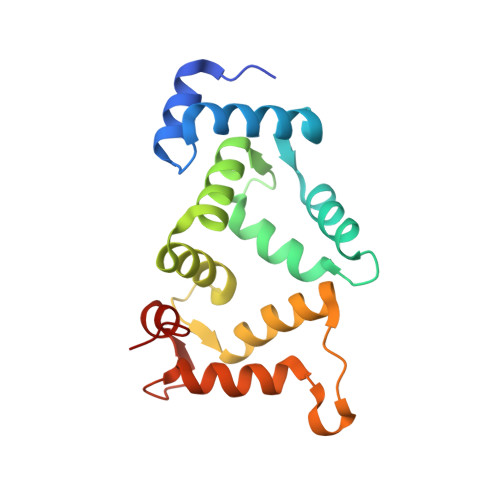
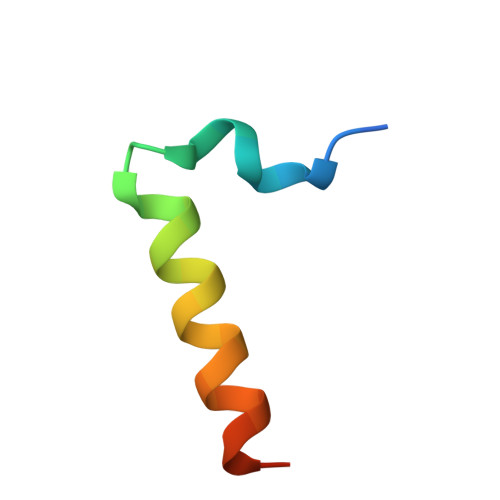
The neuronal calcium sensor NCS-1 regulates the phosphorylation state and activity of the G alpha chaperone and GEF Ric-8A.
Munoz-Reyes, D., McClelland, L.J., Arroyo-Urea, S., Sanchez-Yepes, S., Sabin, J., Perez-Suarez, S., Menendez, M., Mansilla, A., Garcia-Nafria, J., Sprang, S., Sanchez-Barrena, M.J.(2023) Elife 12
- PubMed: 38018500 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.7554/eLife.86151
- Primary Citation Related Structures:
8AHY, 8ALH, 8ALM - PubMed Abstract:
The neuronal calcium sensor 1 (NCS-1), an EF-hand Ca 2+ binding protein, and Ric-8A coregulate synapse number and probability of neurotransmitter release. Recently, the structures of Ric-8A bound to Gα have revealed how Ric-8A phosphorylation promotes Gα recognition and activity as a chaperone and guanine nucleotide exchange factor. However, the molecular mechanism by which NCS-1 regulates Ric-8A activity and its interaction with Gα subunits is not well understood. Given the interest in the NCS-1/Ric-8A complex as a therapeutic target in nervous system disorders, it is necessary to shed light on this molecular mechanism of action at atomic level. We have reconstituted NCS-1/Ric-8A complexes to conduct a multimodal approach and determine the sequence of Ca 2+ signals and phosphorylation events that promote the interaction of Ric-8A with Gα. Our data show that the binding of NCS-1 and Gα to Ric-8A are mutually exclusive. Importantly, NCS-1 induces a structural rearrangement in Ric-8A that traps the protein in a conformational state that is inaccessible to casein kinase II-mediated phosphorylation, demonstrating one aspect of its negative regulation of Ric-8A-mediated G-protein signaling. Functional experiments indicate a loss of Ric-8A guanine nucleotide exchange factor (GEF) activity toward Gα when complexed with NCS-1, and restoration of nucleotide exchange activity upon increasing Ca 2+ concentration. Finally, the high-resolution crystallographic data reported here define the NCS-1/Ric-8A interface and will allow the development of therapeutic synapse function regulators with improved activity and selectivity.
- Department of Crystallography and Structural Biology, Institute of Physical-Chemistry 'Blas Cabrera', CSIC, Madrid, Spain.
Organizational Affiliation: