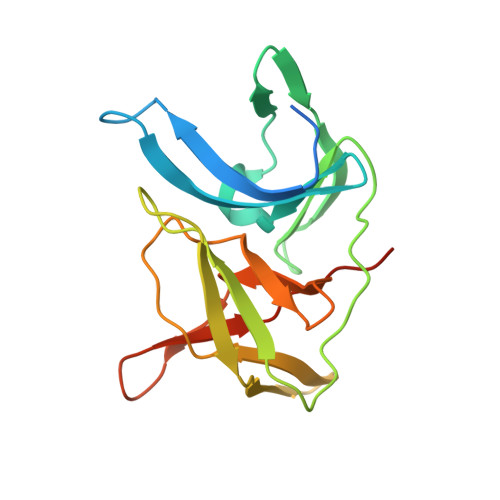
Boroleucine-Derived Covalent Inhibitors of the ZIKV Protease.
Braun, N.J., Huber, S., Schmacke, L.C., Heine, A., Steinmetzer, T.(2023) ChemMedChem 18: e202200336-e202200336
- PubMed: 36325810
- DOI: https://doi.org/10.1002/cmdc.202200336
- Primary Citation Related Structures:
7ZNO - PubMed Abstract:
The Zika virus (ZIKV) remains a potential threat to the public health due to the lack of both an approved vaccination or a specific treatment. In this work, a series of peptidic inhibitors of the ZIKV protease with boroleucine as P1 residue was synthesized. The highest affinities with K i values down to 8 nM were observed for compounds with basic residues in both P2 and P3 position and at the N-terminus. The low potency of reference compounds containing leucine, leucine-amide or isopentylamide as P1 residue suggested a covalent binding mode of the boroleucine-derived inhibitors. This was finally proven by crystal structure determination of the most potent inhibitor from this series in complex with the ZIKV protease.
- Institute of Pharmaceutical Chemistry, Philipps University of Marburg, Marbacher Weg 6, 35032, Marburg, Germany.
Organizational Affiliation: