A quinolin-8-ol sub-millimolar inhibitor of UGGT, the ER glycoprotein quality control checkpoint
Caputo, A.T., Ibba, R., Le Cornu, J.D., Darlot, B., Hensen, M., Lipp, C.B., Marciano, G., Vasiljevic, S., Roversi, P., Zitzmann, N.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| UDP-glucose-glycoprotein glucosyltransferase-like protein | 299 | Thermochaetoides thermophila | Mutation(s): 0 Gene Names: CTHT_0048990 | 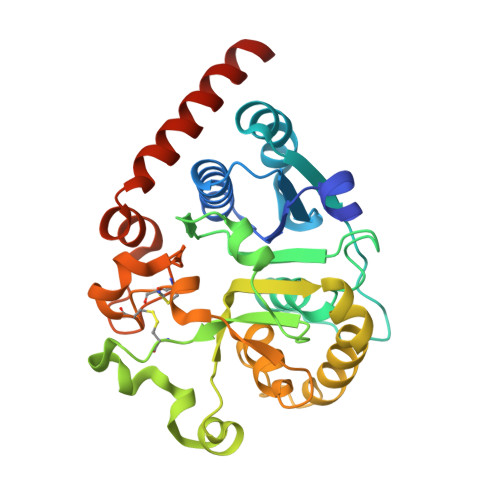 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | G0SB58 | ||||
Glycosylation | |||||
| Glycosylation Sites: 1 | |||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| UPG (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | C [auth A] | URIDINE-5'-DIPHOSPHATE-GLUCOSE C15 H24 N2 O17 P2 HSCJRCZFDFQWRP-JZMIEXBBSA-N |  | ||
| PDO Download:Ideal Coordinates CCD File | H [auth A], I [auth A], J [auth A] | 1,3-PROPANDIOL C3 H8 O2 YPFDHNVEDLHUCE-UHFFFAOYSA-N |  | ||
| IPA Download:Ideal Coordinates CCD File | E [auth A], F [auth A], G [auth A] | ISOPROPYL ALCOHOL C3 H8 O KFZMGEQAYNKOFK-UHFFFAOYSA-N |  | ||
| CA (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | D [auth A] | CALCIUM ION Ca BHPQYMZQTOCNFJ-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 121.442 | α = 90 |
| b = 121.442 | β = 90 |
| c = 62.117 | γ = 120 |
| Software Name | Purpose |
|---|---|
| BUSTER | refinement |
| XDS | data reduction |
| Aimless | data scaling |
| autoBUSTER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Wellcome Trust | United Kingdom | 106272/Z/14/Z |
| Wellcome Trust | United Kingdom | 204801/Z/16/Z |
| Wellcome Trust | United Kingdom | 214090/Z/18/Z |