Aspartyl/Asparaginyl beta-hydroxylase (AspH) oxygenase and TPR domains in complex with D-4-hydroxy-2-oxoglutarate and factor X-derived peptide (39mer-4Ser)
Nakashima, Y., Brewitz, L., Schofield, C.J.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Aspartyl/asparaginyl beta-hydroxylase | 429 | Homo sapiens | Mutation(s): 0 Gene Names: ASPH EC: 1.14.11.16 | 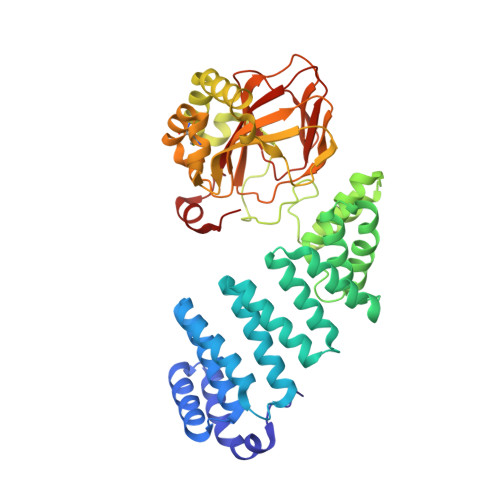 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q12797 GTEx: ENSG00000198363 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q12797 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Coagulation factor X | 39 | synthetic construct | Mutation(s): 4 Gene Names: F10 EC: 3.4.21.6 | 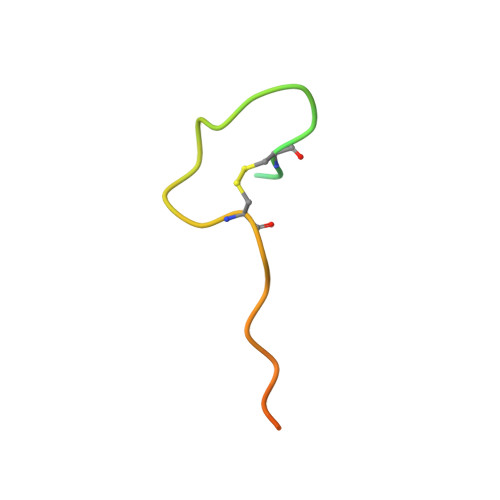 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P00742 GTEx: ENSG00000126218 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P00742 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| IO9 (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | D [auth A] | (2~{R})-2-oxidanyl-4-oxidanylidene-pentanedioic acid C5 H6 O6 WXSKVKPSMAHCSG-UWTATZPHSA-N |  | ||
| MN Download:Ideal Coordinates CCD File | C [auth A] | MANGANESE (II) ION Mn WAEMQWOKJMHJLA-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 87.457 | α = 90 |
| b = 123.608 | β = 90 |
| c = 50.183 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| autoPROC | data reduction |
| autoPROC | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Medical Research Council (MRC, United Kingdom) | United Kingdom | -- |