Structural Basis for Nav1.5 Opening Modulated by a Gating Modifier Toxin
Jiang, D., Catterall, W.A.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Sodium channel protein type 5 subunit alpha,G protein/GFP fusion protein | 1,838 | Rattus norvegicus, Recombinant vesicular stomatitis Indiana virus rVSV-G/GFP | Mutation(s): 0 Gene Names: Scn5a, G, GFP Membrane Entity: Yes | 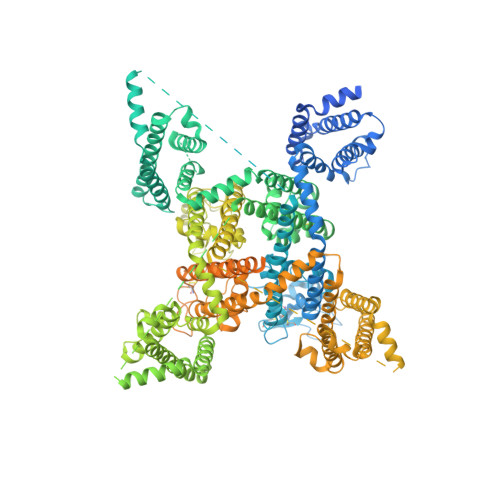 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Groups | P15389B7UCZ6 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Alpha-like toxin Lqh3 | 67 | Leiurus hebraeus | Mutation(s): 0 | 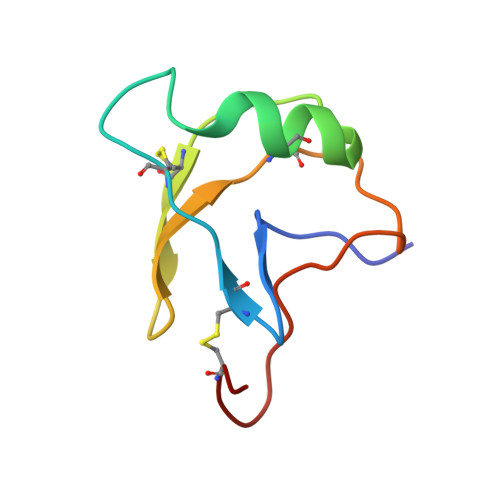 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P56678 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| 6OU Download:Ideal Coordinates CCD File | I [auth A] J [auth A] K [auth A] L [auth A] M [auth A] | [(2~{R})-1-[2-azanylethoxy(oxidanyl)phosphoryl]oxy-3-hexadecanoyloxy-propan-2-yl] (~{Z})-octadec-9-enoate C39 H76 N O8 P FHQVHHIBKUMWTI-OTMQOFQLSA-N |  | ||
| 9Z9 Download:Ideal Coordinates CCD File | O [auth A] | (3beta,14beta,17beta,25R)-3-[4-methoxy-3-(methoxymethyl)butoxy]spirost-5-en C34 H56 O5 CEEBZAXXSRFQIC-GZSGZGDASA-N |  | ||
| NAG Download:Ideal Coordinates CCD File | C [auth A], D [auth A], E [auth A], F [auth A], G [auth A] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
| BMA Download:Ideal Coordinates CCD File | H [auth A] | beta-D-mannopyranose C6 H12 O6 WQZGKKKJIJFFOK-RWOPYEJCSA-N |  | ||
| Task | Software Package | Version |
|---|---|---|
| MODEL REFINEMENT | PHENIX | |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Heart, Lung, and Blood Institute (NIH/NHLBI) | United States | -- |