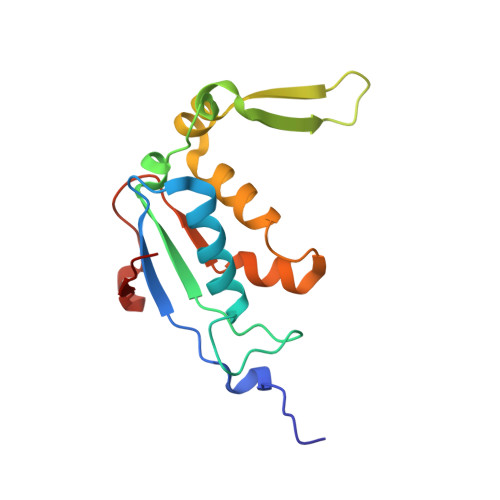
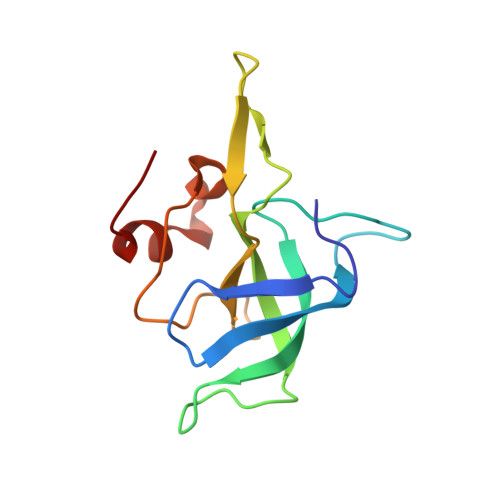
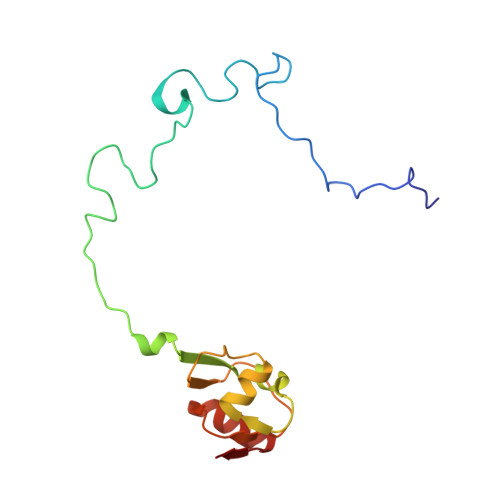
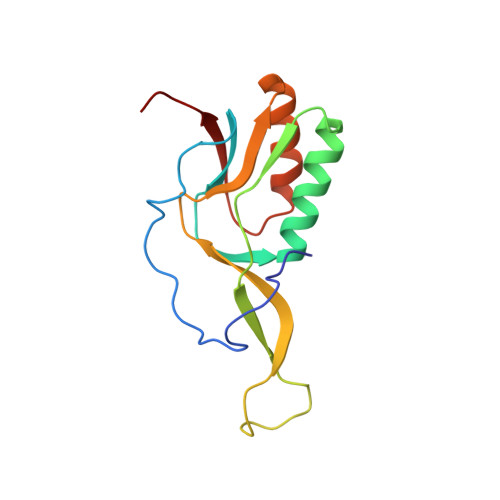
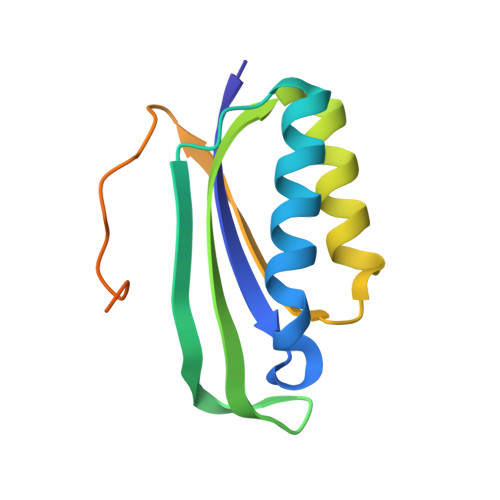
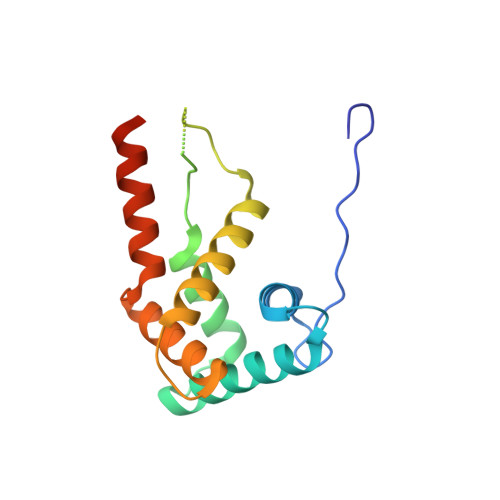
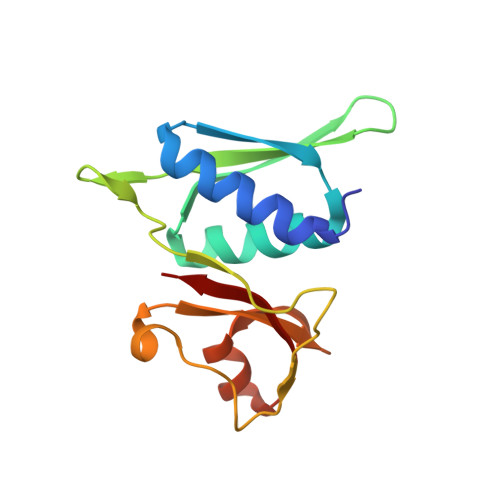
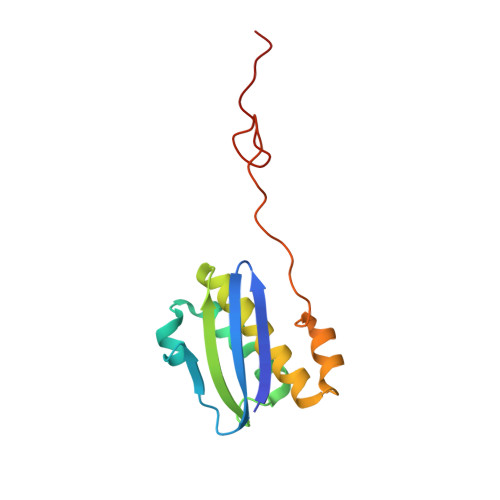
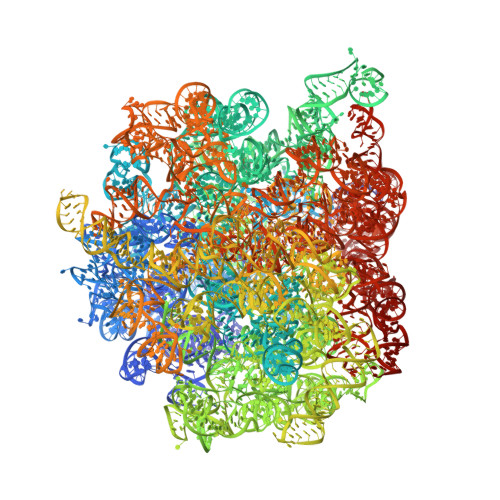
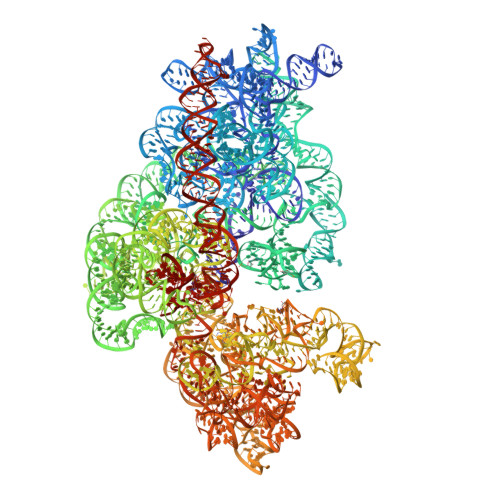
Streptothricin F is a bactericidal antibiotic effective against highly drug-resistant gram-negative bacteria that interacts with the 30S subunit of the 70S ribosome.
Morgan, C.E., Kang, Y.S., Green, A.B., Smith, K.P., Dowgiallo, M.G., Miller, B.C., Chiaraviglio, L., Truelson, K.A., Zulauf, K.E., Rodriguez, S., Kang, A.D., Manetsch, R., Yu, E.W., Kirby, J.E.(2023) PLoS Biol 21: e3002091-e3002091
- PubMed: 37192172 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1371/journal.pbio.3002091
- Primary Citation Related Structures:
7UVV, 7UVW, 7UVX, 7UVY, 7UVZ, 7UW1 - PubMed Abstract:
The streptothricin natural product mixture (also known as nourseothricin) was discovered in the early 1940s, generating intense initial interest because of excellent gram-negative activity. Here, we establish the activity spectrum of nourseothricin and its main components, streptothricin F (S-F, 1 lysine) and streptothricin D (S-D, 3 lysines), purified to homogeneity, against highly drug-resistant, carbapenem-resistant Enterobacterales (CRE) and Acinetobacter baumannii. For CRE, the MIC50 and MIC90 for S-F and S-D were 2 and 4 μM, and 0.25 and 0.5 μM, respectively. S-F and nourseothricin showed rapid, bactericidal activity. S-F and S-D both showed approximately 40-fold greater selectivity for prokaryotic than eukaryotic ribosomes in in vitro translation assays. In vivo, delayed renal toxicity occurred at >10-fold higher doses of S-F compared with S-D. Substantial treatment effect of S-F in the murine thigh model was observed against the otherwise pandrug-resistant, NDM-1-expressing Klebsiella pneumoniae Nevada strain with minimal or no toxicity. Cryo-EM characterization of S-F bound to the A. baumannii 70S ribosome defines extensive hydrogen bonding of the S-F steptolidine moiety, as a guanine mimetic, to the 16S rRNA C1054 nucleobase (Escherichia coli numbering) in helix 34, and the carbamoylated gulosamine moiety of S-F with A1196, explaining the high-level resistance conferred by corresponding mutations at the residues identified in single rrn operon E. coli. Structural analysis suggests that S-F probes the A-decoding site, which potentially may account for its miscoding activity. Based on unique and promising activity, we suggest that the streptothricin scaffold deserves further preclinical exploration as a potential therapeutic for drug-resistant, gram-negative pathogens.
- Department of Pharmacology, Case Western Reserve University School of Medicine, Cleveland, Ohio, United States of America.
Organizational Affiliation: