Mechanistic plasticity in ApmA enables aminoglycoside promiscuity for resistance
Bordeleau, E., Stogios, P.J., Evdokimova, E., Koteva, K., Savchenko, A., Wright, G.D.(2024) Nat Chem Biol 20: 234-242
Experimental Data Snapshot
Starting Model: experimental
View more details
(2024) Nat Chem Biol 20: 234-242
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Aminocyclitol acetyltransferase ApmA | 276 | Staphylococcus aureus | Mutation(s): 0 Gene Names: apmA | 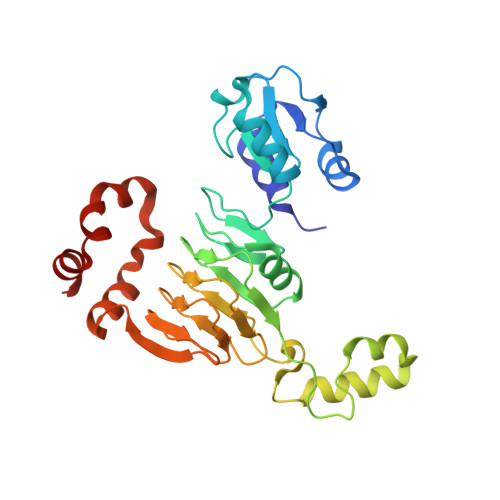 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A1D0AST6 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 5 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| COA (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | AA [auth D], FA [auth E], H [auth A], M [auth B], T [auth C] | COENZYME A C21 H36 N7 O16 P3 S RGJOEKWQDUBAIZ-IBOSZNHHSA-N |  | ||
| 9CS (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | EA [auth E] G [auth A] L [auth B] R [auth C] S [auth C] | (1R,2S,3S,4R,6S)-4,6-DIAMINO-3-[(3-AMINO-3-DEOXY-ALPHA-D-GLUCOPYRANOSYL)OXY]-2-HYDROXYCYCLOHEXYL 2,6-DIAMINO-2,6-DIDEOXY-ALPHA-D-GLUCOPYRANOSIDE C18 H37 N5 O10 SKKLOUVUUNMCJE-FQSMHNGLSA-N |  | ||
| SO4 Download:Ideal Coordinates CCD File | CA [auth D] HA [auth E] J [auth A] K [auth A] KA [auth F] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| EDO Download:Ideal Coordinates CCD File | BA [auth D] GA [auth E] I [auth A] IA [auth F] JA [auth F] | 1,2-ETHANEDIOL C2 H6 O2 LYCAIKOWRPUZTN-UHFFFAOYSA-N |  | ||
| CL Download:Ideal Coordinates CCD File | DA [auth D] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 101.093 | α = 90 |
| b = 131.685 | β = 90 |
| c = 157.915 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| HKL-3000 | data reduction |
| HKL-3000 | data scaling |
| PHENIX | phasing |
| PHENIX | model building |
| Coot | model building |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute Of Allergy and Infectious Diseases (NIH/NIAID) | United States | HHSN272201700060C |