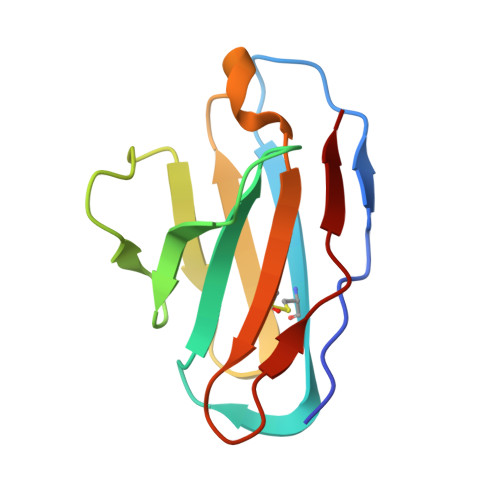
A cocktail of protective antibodies subverts the dense glycan shield of Lassa virus.
Li, H., Buck, T., Zandonatti, M., Yin, J., Moon-Walker, A., Fang, J., Koval, A., Heinrich, M.L., Rowland, M.M., Diaz Avalos, R., Schendel, S.L., Parekh, D., Zyla, D., Enriquez, A., Harkins, S., Sullivan, B., Smith, V., Chukwudozie, O., Watanabe, R., Robinson, J.E., Garry, R.F., Branco, L.M., Hastie, K.M., Saphire, E.O.(2022) Sci Transl Med 14: eabq0991-eabq0991
- PubMed: 36288283 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1126/scitranslmed.abq0991
- Primary Citation Related Structures:
7UOT, 7UOV - PubMed Abstract:
Developing potent therapeutics and effective vaccines are the ultimate goals in controlling infectious diseases. Lassa virus (LASV), the causative pathogen of Lassa fever (LF), infects hundreds of thousands annually, but effective antivirals or vaccines against LASV infection are still lacking. Furthermore, neutralizing antibodies against LASV are rare. Here, we describe biochemical analyses and high-resolution cryo-electron microscopy structures of a therapeutic cocktail of three broadly protective antibodies that target the LASV glycoprotein complex (GPC), previously identified from survivors of multiple LASV infections. Structural and mechanistic analyses reveal compatible neutralizing epitopes and complementary neutralization mechanisms that offer high potency, broad range, and resistance to escape. These antibodies either circumvent or exploit specific glycans comprising the extensive glycan shield of GPC. Further, they require mammalian glycosylation, native GPC cleavage, and proper GPC trimerization. These findings guided engineering of a next-generation GPC antigen suitable for future neutralizing antibody and vaccine discovery. Together, these results explain protective mechanisms of rare, broad, and potent antibodies and identify a strategy for the rational design of therapeutic modalities against LF and related infectious diseases.
- Center for Infectious Disease and Vaccine Research, La Jolla Institute for Immunology, 9420 Athena Circle, La Jolla, CA 92037, USA.
Organizational Affiliation: