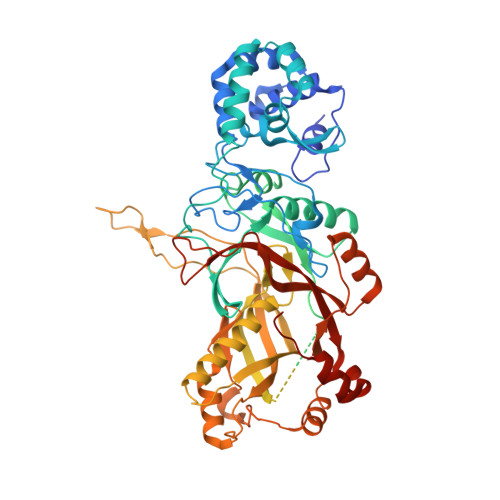
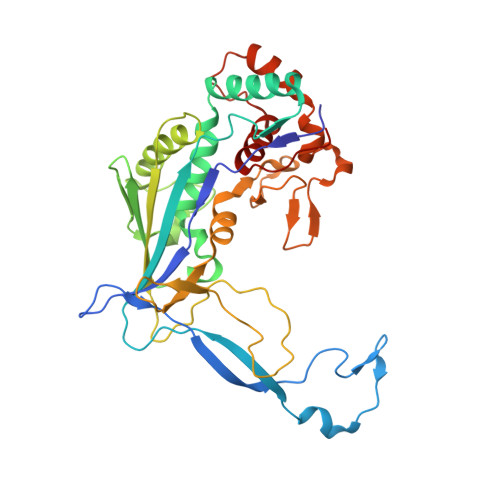
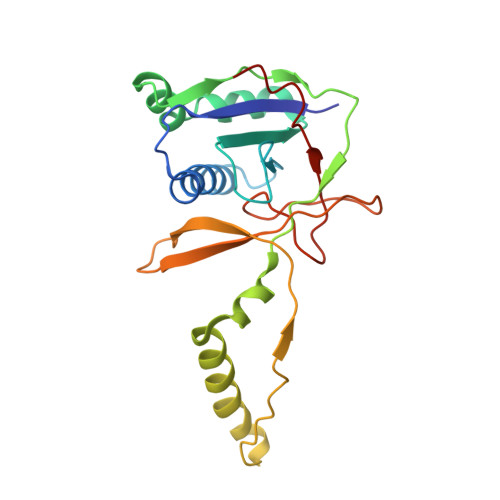
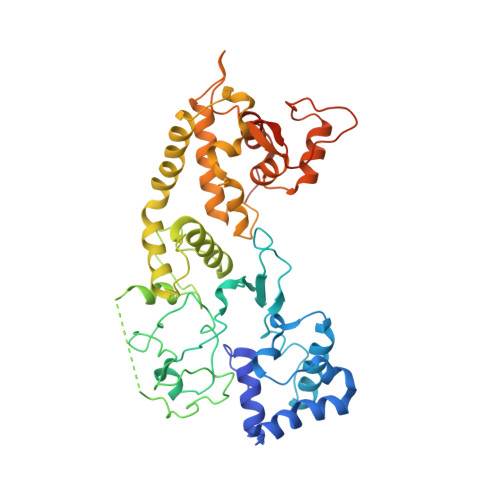
Multiple adaptations underly co-option of a CRISPR surveillance complex for RNA-guided DNA transposition.
Park, J.U., Petassi, M.T., Hsieh, S.C., Mehrotra, E., Schuler, G., Budhathoki, J., Truong, V.H., Thyme, S.B., Ke, A., Kellogg, E.H., Peters, J.E.(2023) Mol Cell 83: 1827-1838.e6
- PubMed: 37267904 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.molcel.2023.05.005
- Primary Citation Related Structures:
7U5D, 7U5E - PubMed Abstract:
CRISPR-associated transposons (CASTs) are natural RNA-directed transposition systems. We demonstrate that transposon protein TniQ plays a central role in promoting R-loop formation by RNA-guided DNA-targeting modules. TniQ residues, proximal to CRISPR RNA (crRNA), are required for recognizing different crRNA categories, revealing an unappreciated role of TniQ to direct transposition into different classes of crRNA targets. To investigate adaptations allowing CAST elements to utilize attachment sites inaccessible to CRISPR-Cas surveillance complexes, we compared and contrasted PAM sequence requirements in both I-F3b CAST and I-F1 CRISPR-Cas systems. We identify specific amino acids that enable a wider range of PAM sequences to be accommodated in I-F3b CAST elements compared with I-F1 CRISPR-Cas, enabling CAST elements to access attachment sites as sequences drift and evade host surveillance. Together, this evidence points to the central role of TniQ in facilitating the acquisition of CRISPR effector complexes for RNA-guided DNA transposition.
- Department of Molecular Biology and Genetics, Cornell University, Ithaca, NY 14853, USA.
Organizational Affiliation: