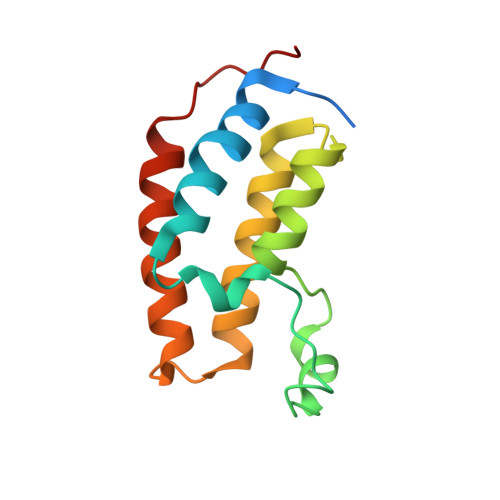
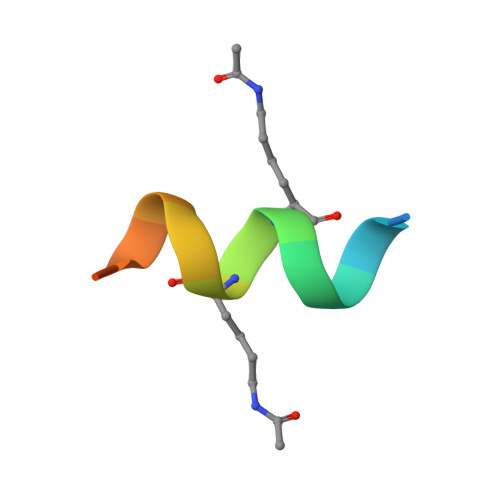
mRNA display reveals a class of high-affinity bromodomain-binding motifs that are not found in the human proteome.
Low, J.K.K., Patel, K., Jones, N., Solomon, P., Norman, A., Maxwell, J.W.C., Pachl, P., Matthews, J.M., Payne, R.J., Passioura, T., Suga, H., Walport, L.J., Mackay, J.P.(2023) J Biological Chem 299: 105482-105482
- PubMed: 37992806 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jbc.2023.105482
- Primary Citation Related Structures:
7TO7, 7TO8, 7TO9, 7TOA - PubMed Abstract:
Bromodomains (BDs) regulate gene expression by recognizing protein motifs containing acetyllysine. Although originally characterized as histone-binding proteins, it has since become clear that these domains interact with other acetylated proteins, perhaps most prominently transcription factors. The likely transient nature and low stoichiometry of such modifications, however, has made it challenging to fully define the interactome of any given BD. To begin to address this knowledge gap in an unbiased manner, we carried out mRNA display screens against a BD-the N-terminal BD of BRD3-using peptide libraries that contained either one or two acetyllysine residues. We discovered peptides with very strong consensus sequences and with affinities that are significantly higher than typical BD-peptide interactions. X-ray crystal structures also revealed modes of binding that have not been seen with natural ligands. Intriguingly, however, our sequences are not found in the human proteome, perhaps suggesting that strong binders to BDs might have been selected against during evolution.
- School of Life and Environmental Sciences, University of Sydney, New South Wales, Australia.
Organizational Affiliation: