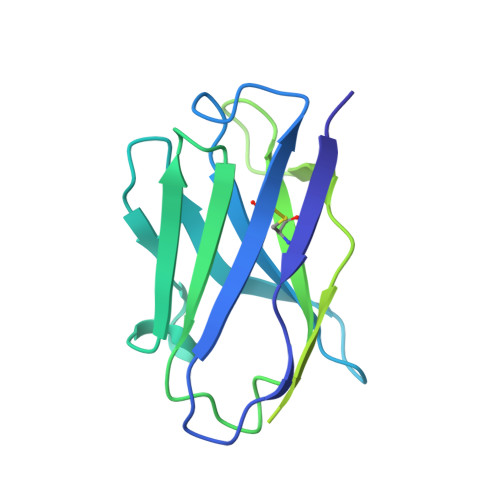
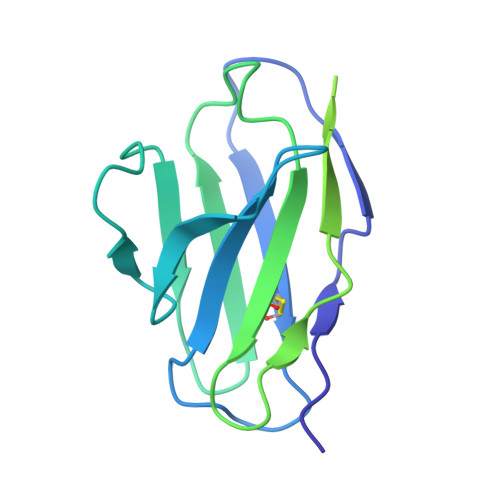
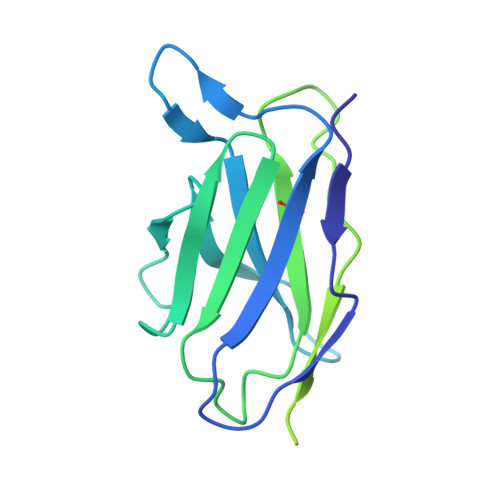
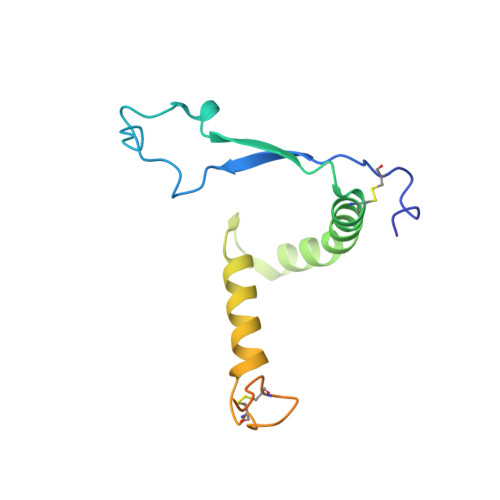
Structure of the Inmazeb cocktail and resistance to Ebola virus escape.
Rayaprolu, V., Fulton, B.O., Rafique, A., Arturo, E., Williams, D., Hariharan, C., Callaway, H., Parvate, A., Schendel, S.L., Parekh, D., Hui, S., Shaffer, K., Pascal, K.E., Wloga, E., Giordano, S., Negron, N., Ni, M., Copin, R., Atwal, G.S., Franklin, M., Boytz, R.M., Donahue, C., Davey, R., Baum, A., Kyratsous, C.A., Saphire, E.O.(2023) Cell Host Microbe 31: 260
- PubMed: 36708708 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.chom.2023.01.002
- Primary Citation Related Structures:
7TN9 - PubMed Abstract:
Monoclonal antibodies can provide important pre- or post-exposure protection against infectious disease for those not yet vaccinated or in individuals that fail to mount a protective immune response after vaccination. Inmazeb (REGN-EB3), a three-antibody cocktail against Ebola virus, lessened disease and improved survival in a controlled trial. Here, we present the cryo-EM structure at 3.1 Å of the Ebola virus glycoprotein, determined without symmetry averaging, in a simultaneous complex with the antibodies in the Inmazeb cocktail. This structure allows the modeling of previously disordered portions of the glycoprotein glycan cap, maps the non-overlapping epitopes of Inmazeb, and illuminates the basis for complementary activities and residues critical for resistance to escape by these and other clinically relevant antibodies. We further provide direct evidence that Inmazeb protects against the rapid emergence of escape mutants, whereas monotherapies even against conserved epitopes do not, supporting the benefit of a cocktail versus a monotherapy approach.
- La Jolla Institute for Immunology, La Jolla, CA 92037, USA.
Organizational Affiliation: