A crystallography-based study of fragment extensions into the 14-3-3 binding groove
Centorrino, F., Wu, Q., Cossar, P., Brunsveld, L., Ottmann, C.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| 14-3-3 protein sigma | 253 | Homo sapiens | Mutation(s): 0 Gene Names: SFN, HME1 | 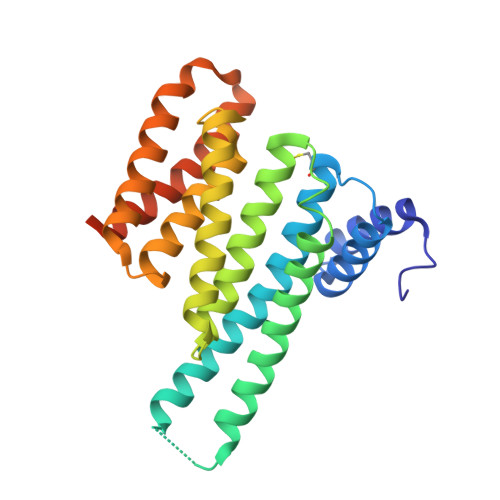 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P31947 GTEx: ENSG00000175793 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P31947 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cyclin-dependent kinase inhibitor 1B | B [auth P] | 12 | Homo sapiens | Mutation(s): 0 | 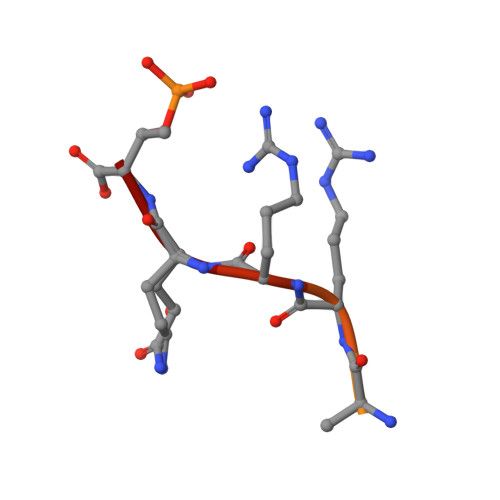 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P46527 GTEx: ENSG00000111276 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P46527 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| 0AW (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | C [auth A], D [auth A], E [auth A] | ~{N}-[(5-carbamimidoyl-3-phenyl-thiophen-2-yl)methyl]-2,3-dihydro-1-benzofuran-5-carboxamide C21 H19 N3 O2 S OKYRJCCVSZQTDE-UHFFFAOYSA-N |  | ||
| CA Download:Ideal Coordinates CCD File | I [auth A] | CALCIUM ION Ca BHPQYMZQTOCNFJ-UHFFFAOYSA-N |  | ||
| CL Download:Ideal Coordinates CCD File | G [auth A] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| MG Download:Ideal Coordinates CCD File | F [auth A], H [auth A] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Modified Residues 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| CSO Query on CSO | A | L-PEPTIDE LINKING | C3 H7 N O3 S |  | CYS |
| TPO Query on TPO | B [auth P] | L-PEPTIDE LINKING | C4 H10 N O6 P |  | THR |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 82.55 | α = 90 |
| b = 111.558 | β = 90 |
| c = 62.346 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| DIALS | data reduction |
| Aimless | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| H2020 Marie Curie Actions of the European Commission | European Union | 675179 |