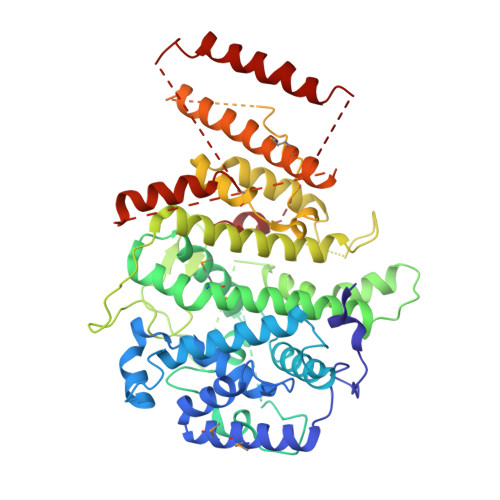
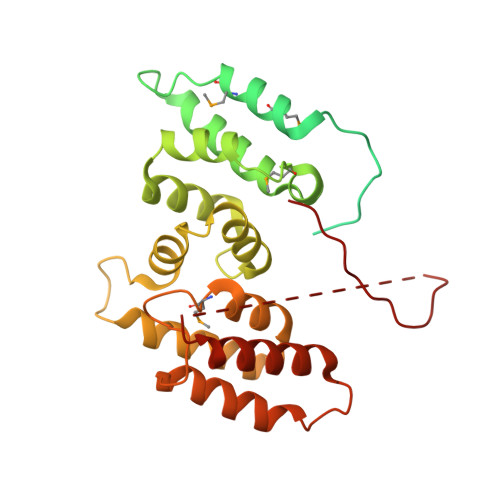
Nse5/6 inhibits the Smc5/6 ATPase and modulates DNA substrate binding.
Taschner, M., Basquin, J., Steigenberger, B., Schafer, I.B., Soh, Y.M., Basquin, C., Lorentzen, E., Raschle, M., Scheltema, R.A., Gruber, S.(2021) EMBO J 40: e107807-e107807
- PubMed: 34191293 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.15252/embj.2021107807
- Primary Citation Related Structures:
7OGG - PubMed Abstract:
Eukaryotic cells employ three SMC (structural maintenance of chromosomes) complexes to control DNA folding and topology. The Smc5/6 complex plays roles in DNA repair and in preventing the accumulation of deleterious DNA junctions. To elucidate how specific features of Smc5/6 govern these functions, we reconstituted the yeast holo-complex. We found that the Nse5/6 sub-complex strongly inhibited the Smc5/6 ATPase by preventing productive ATP binding. This inhibition was relieved by plasmid DNA binding but not by short linear DNA, while opposing effects were observed without Nse5/6. We uncovered two binding sites for Nse5/6 on Smc5/6, based on an Nse5/6 crystal structure and cross-linking mass spectrometry data. One binding site is located at the Smc5/6 arms and one at the heads, the latter likely exerting inhibitory effects on ATP hydrolysis. Cysteine cross-linking demonstrated that the interaction with Nse5/6 anchored the ATPase domains in a non-productive state, which was destabilized by ATP and DNA. Under similar conditions, the Nse4/3/1 module detached from the ATPase. Altogether, we show how DNA substrate selection is modulated by direct inhibition of the Smc5/6 ATPase by Nse5/6.
- Department of Fundamental Microbiology (DMF), Faculty of Biology and Medicine (FBM), University of Lausanne (UNIL), Lausanne, Switzerland.
Organizational Affiliation: