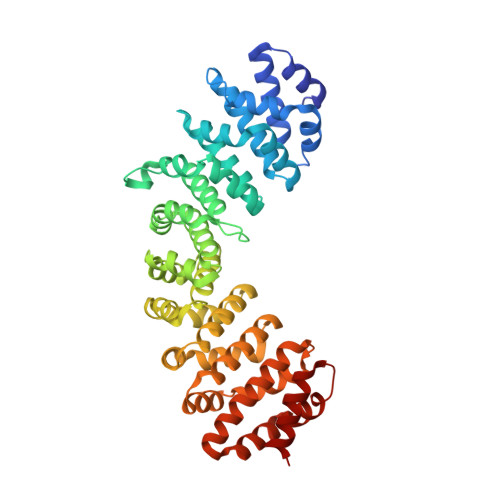
Differential recognition of canonical NF-kappa B dimers by Importin alpha 3.
Florio, T.J., Lokareddy, R.K., Yeggoni, D.P., Sankhala, R.S., Ott, C.A., Gillilan, R.E., Cingolani, G.(2022) Nat Commun 13: 1207-1207
- PubMed: 35260573 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-022-28846-z
- Primary Citation Related Structures:
7LEQ, 7LET, 7LEU, 7LF4, 7LFC - PubMed Abstract:
Nuclear translocation of the p50/p65 heterodimer is essential for NF-κB signaling. In unstimulated cells, p50/p65 is retained by the inhibitor IκBα in the cytoplasm that masks the p65-nuclear localization sequence (NLS). Upon activation, p50/p65 is translocated into the nucleus by the adapter importin α3 and the receptor importin β. Here, we describe a bipartite NLS in p50/p65, analogous to nucleoplasmin NLS but exposed in trans. Importin α3 accommodates the p50- and p65-NLSs at the major and minor NLS-binding pockets, respectively. The p50-NLS is the predominant binding determinant, while the p65-NLS induces a conformational change in the Armadillo 7 of importin α3 that stabilizes a helical conformation of the p65-NLS. Neither conformational change was observed for importin α1, which makes fewer bonds with the p50/p65 NLSs, explaining the preference for α3. We propose that importin α3 discriminates between the transcriptionally active p50/p65 heterodimer and p50/p50 and p65/65 homodimers, ensuring fidelity in NF-κB signaling.
- Department of Biochemistry and Molecular Biology, Thomas Jefferson University, 1020 Locust Street, Philadelphia, PA, 19107, USA.
Organizational Affiliation: