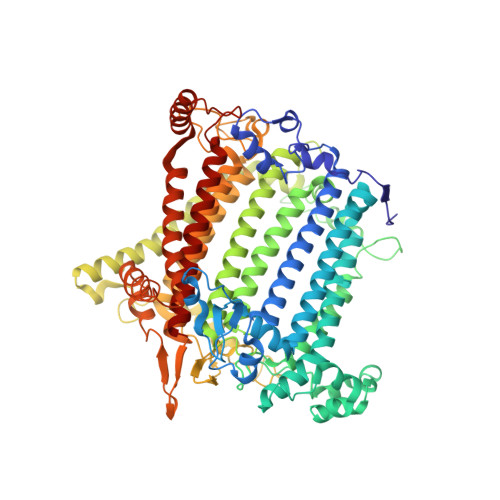
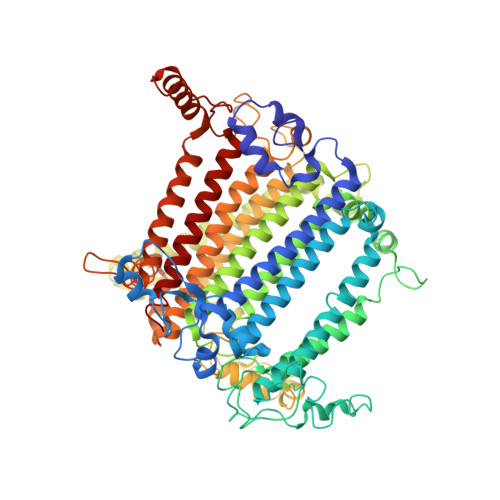
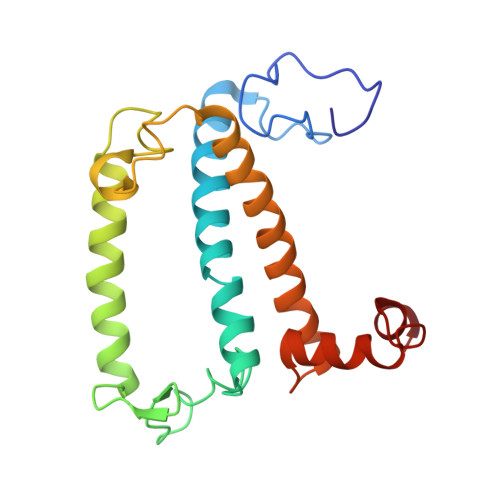
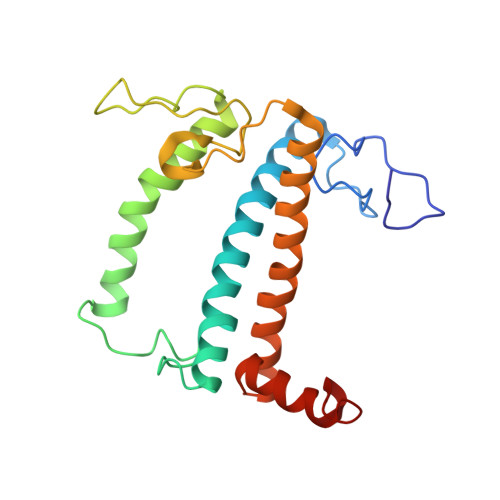
The structure of the Physcomitrium patens photosystem I reveals a unique Lhca2 paralogue replacing Lhca4.
Gorski, C., Riddle, R., Toporik, H., Da, Z., Dobson, Z., Williams, D., Mazor, Y.(2022) Nat Plants 8: 307-316
- PubMed: 35190662 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41477-022-01099-w
- Primary Citation Related Structures:
7KSQ, 7KU5, 7KUX - PubMed Abstract:
The moss Physcomitrium patens diverged from green algae shortly after the colonization of land by ancient plants. This colonization posed new environmental challenges, which drove evolutionary processes. The photosynthetic machinery of modern flowering plants is adapted to the high light conditions on land. Red-shifted Lhca4 antennae are present in the photosystem I light-harvesting complex of many green-lineage plants but absent in P. patens. The cryo-EM structure of the P. patens photosystem I light-harvesting complex I supercomplex (PSI-LHCI) at 2.8 Å reveals that Lhca4 is replaced by a unique Lhca2 paralogue in moss. This PSI-LHCI supercomplex also retains the PsaM subunit, present in Cyanobacteria and several algal species but lost in vascular plants, and the PsaO subunit responsible for binding light-harvesting complex II. The blue-shifted Lhca2 paralogue and chlorophyll b enrichment relative to flowering plants make the P. patens PSI-LHCI spectroscopically unique among other green-lineage supercomplexes. Overall, the structure represents an evolutionary intermediate PSI with the crescent-shaped LHCI common in vascular plants, and contains a unique Lhca2 paralogue that facilitates the moss's adaptation to low-light niches.
- School of Molecular Sciences, Arizona State University, Tempe, AZ, USA.
Organizational Affiliation: