Inhibition of the angiotensin II type 2 receptor AT 2 R is a novel therapeutic strategy for glioblastoma.
Perryman, R., Renziehausen, A., Shaye, H., Kostagianni, A.D., Tsiailanis, A.D., Thorne, T., Chatziathanasiadou, M.V., Sivolapenko, G.B., El Mubarak, M.A., Han, G.W., Zarzycka, B., Katritch, V., Lebon, G., Lo Nigro, C., Lattanzio, L., Morse, S.V., Choi, J.J., O'Neill, K., Kanaki, Z., Klinakis, A., Crook, T., Cherezov, V., Tzakos, A.G., Syed, N.(2022) Proc Natl Acad Sci U S A 119: e2116289119-e2116289119
- PubMed: 35917342 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.2116289119
- Primary Citation Related Structures:
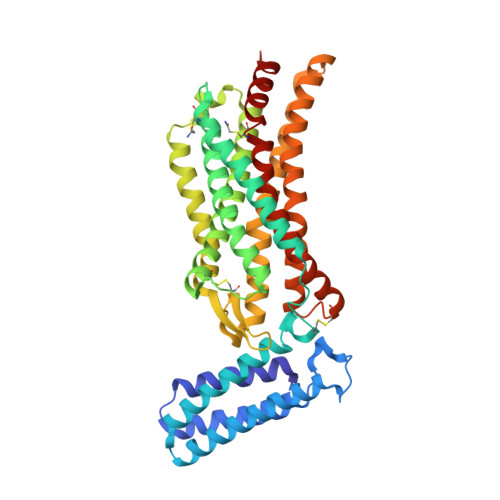
7JNI - PubMed Abstract:
Glioblastoma (GBM) is an aggressive malignant primary brain tumor with limited therapeutic options. We show that the angiotensin II (AngII) type 2 receptor (AT 2 R) is a therapeutic target for GBM and that AngII, endogenously produced in GBM cells, promotes proliferation through AT 2 R. We repurposed EMA401, an AT 2 R antagonist originally developed as a peripherally restricted analgesic, for GBM and showed that it inhibits the proliferation of AT 2 R-expressing GBM spheroids and blocks their invasiveness and angiogenic capacity. The crystal structure of AT 2 R bound to EMA401 was determined and revealed the receptor to be in an active-like conformation with helix-VIII blocking G-protein or β-arrestin recruitment. The architecture and interactions of EMA401 in AT 2 R differ drastically from complexes of AT 2 R with other relevant compounds. To enhance central nervous system (CNS) penetration of EMA401, we exploited the crystal structure to design an angiopep-2-tethered EMA401 derivative, A3E. A3E exhibited enhanced CNS penetration, leading to reduced tumor volume, inhibition of proliferation, and increased levels of apoptosis in an orthotopic xenograft model of GBM.
- John Fulcher Molecular Neuro-Oncology Laboratory, Department Brain Sciences, Imperial College, London, United Kingdom.
Organizational Affiliation: