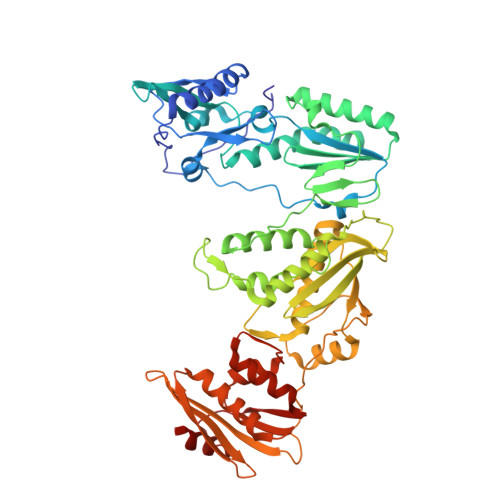
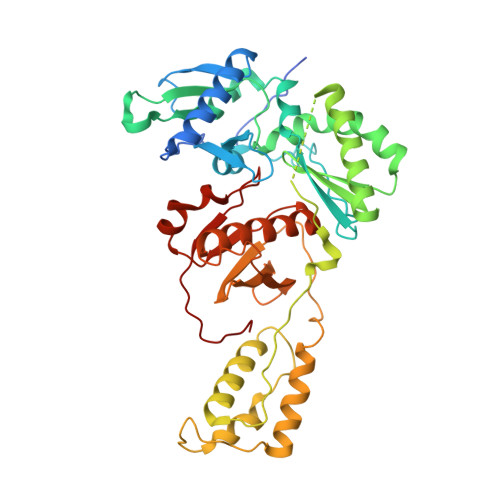
Biochemical and Structural Properties of Entecavir-Resistant Hepatitis B Virus Polymerase with L180M/M204V Mutations.
Nakajima, S., Watashi, K., Kato, T., Muramatsu, M., Wakita, T., Tamura, N., Hattori, S.I., Maeda, K., Mitsuya, H., Yasutake, Y., Toyoda, T.(2021) J Virol 95: e0240120-e0240120
- PubMed: 34076480 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1128/JVI.02401-20
- Primary Citation Related Structures:
7DBM, 7DBN - PubMed Abstract:
Entecavir (ETV) is a widely used anti-hepatitis B virus (HBV) drug. However, the emergence of resistant mutations in HBV reverse transcriptase (RT) results in treatment failure. To understand the mechanism underlying the development of ETV resistance by HBV RT, we analyzed the L180M, M204V, and L180M/M204V mutants using a combination of biochemical and structural techniques. ETV-triphosphate (ETV-TP) exhibited competitive inhibition with dGTP in both wild-type (wt) RT and M204V RT, as observed using Lineweaver-Burk plots. In contrast, RT L180M or L180M/M204V did not fit either competitive, uncompetitive, noncompetitive, or typical mixed inhibition, although ETV-TP was a competitive inhibitor of dGTP. Crystallography of HIV RT Y115F/F116Y/Q151M/F160M/M184V , mimicking HBV RT L180M/M204V, showed that the F115 bulge (F88 in HBV RT) caused by the F160M mutation induced deviated binding of dCTP from its normal tight binding position. Modeling of ETV-TP on the deviated dCTP indicated that a steric clash could occur between ETV-TP methylene and the 3'-end nucleoside ribose. ETV-TP is likely to interact primarily with HBV RT M171 prior to final accommodation at the deoxynucleoside triphosphate (dNTP) binding site (Y. Yasutake, S. Hattori, H. Hayashi, K. Matsuda, et al., Sci Rep 8:1624, 2018, https://doi.org/10.1038/s41598-018-19602-9). Therefore, in HBV RT L180M/M204V, ETV-TP may be stuck at M171, a residue that is conserved in almost all HBV isolates, leading to the strange inhibition pattern observed in the kinetic analysis. Collectively, our results provide novel insights into the mechanism of ETV resistance of HBV RT caused by L180M and M204V mutations. IMPORTANCE HBV infects 257 million people in the world, who suffer from elevated risks of liver cirrhosis and cancer. ETV is one of the most potent anti-HBV drugs, and ETV resistance mutations in HBV RT have been extensively studied. Nevertheless, the mechanisms underlying ETV resistance have remained elusive. We propose an attractive hypothesis to explain ETV resistance and effectiveness using a combination of kinetic and structural analyses. ETV is likely to have an additional interaction site, M171, beside the dNTP pocket of HBV RT; this finding indicates that nucleos(t)ide analogues (NAs) recognizing multiple interaction sites within RT may effectively inhibit the enzyme. Modification of ETV may render it more effective and enable the rational design of efficient NA inhibitors.
- Department of Virology II, National Institute of Infectious Diseases, Tokyo, Japan.
Organizational Affiliation: