CRYSTAL STRUCTURE OF THE HUMAN PRMT5:MEP50 COMPLEX with JNJB44355437
Brown, D., Robinson, C., Carr, K.H., Pande, V.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Protein arginine N-methyltransferase 5 | A [auth AAA] | 645 | Homo sapiens | Mutation(s): 0 Gene Names: PRMT5, HRMT1L5, IBP72, JBP1, SKB1 EC: 2.1.1.320 | 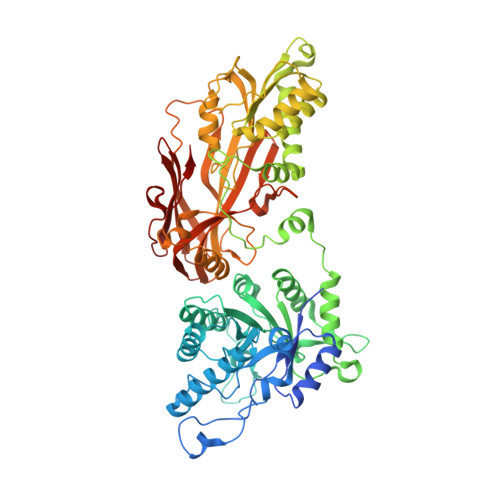 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: O14744 GTEx: ENSG00000100462 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | O14744 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Methylosome protein 50 | B [auth BBB] | 348 | Homo sapiens | Mutation(s): 0 Gene Names: WDR77, MEP50, WD45, HKMT1069, Nbla10071 | 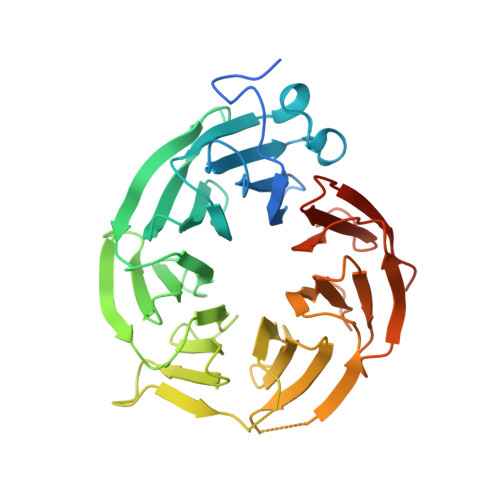 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q9BQA1 GTEx: ENSG00000116455 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9BQA1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| U6K (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | C [auth AAA] | (2~{R},3~{R},4~{S},5~{R})-2-[(4~{a}~{S},7~{a}~{S})-4-azanyl-1,4,4~{a},7~{a}-tetrahydropyrrolo[2,3-d]pyrimidin-7-yl]-5-(quinolin-7-yloxymethyl)oxolane-3,4-diol C20 H23 N5 O4 ZIYBQKLKBCWPQZ-AUENAHFOSA-N |  | ||
| EPE Download:Ideal Coordinates CCD File | D [auth AAA] | 4-(2-HYDROXYETHYL)-1-PIPERAZINE ETHANESULFONIC ACID C8 H18 N2 O4 S JKMHFZQWWAIEOD-UHFFFAOYSA-N |  | ||
| PEG Download:Ideal Coordinates CCD File | H [auth AAA] | DI(HYDROXYETHYL)ETHER C4 H10 O3 MTHSVFCYNBDYFN-UHFFFAOYSA-N |  | ||
| EDO Download:Ideal Coordinates CCD File | E [auth AAA], F [auth AAA], G [auth AAA] | 1,2-ETHANEDIOL C2 H6 O2 LYCAIKOWRPUZTN-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 101.19 | α = 90 |
| b = 137.25 | β = 90 |
| c = 179.37 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| xia2 | data reduction |
| Aimless | data scaling |
| PHASER | phasing |