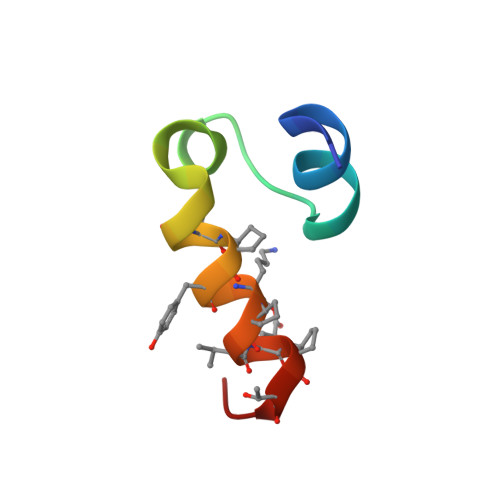A computationally designed beta-amino acid-containing miniprotein.
Bejger, M., Fortuna, P., Drewniak-Switalska, M., Plewka, J., Rypniewski, W., Berlicki, L.(2021) Chem Commun (Camb) 57: 6015-6018
- PubMed: 34032224 Search on PubMed
- DOI: https://doi.org/10.1039/d1cc02192c
- Primary Citation Related Structures:
7ARR, 7ARS - PubMed Abstract:
A new miniprotein built from three helices, including one structure based on the ααβαααβ sequence pattern was developed. Its crystal structure revealed a compact conformation with a well-packed hydrophobic core of unprecedented structure. The miniprotein formed dimers that were stabilized by the interaction of their hydrophobic surfaces.
- Institute of Bioorganic Chemistry, Polish Academy of Sciences, Noskowskiego 12/14, Poznań 61-704, Poland.
Organizational Affiliation: