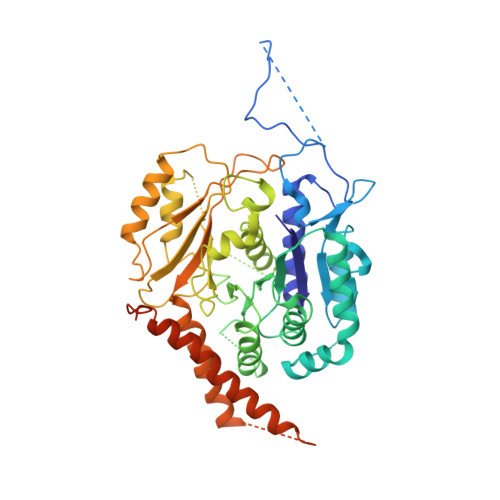
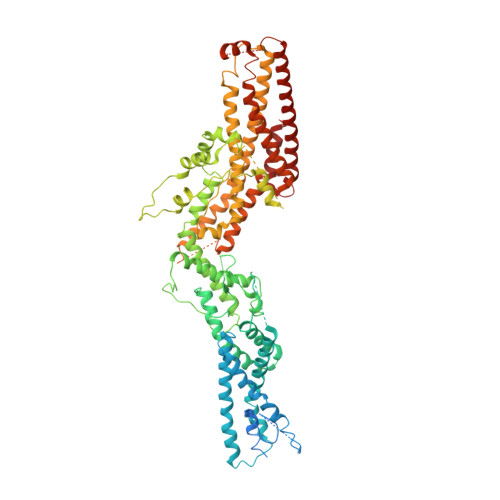
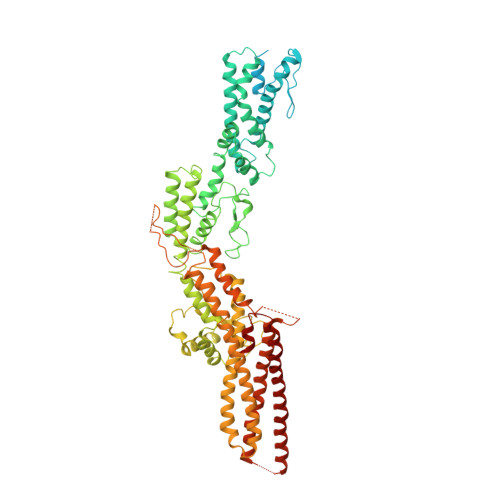
The cryo-EM structure of a gamma-TuSC elucidates architecture and regulation of minimal microtubule nucleation systems.
Zupa, E., Zheng, A., Neuner, A., Wurtz, M., Liu, P., Bohler, A., Schiebel, E., Pfeffer, S.(2020) Nat Commun 11: 5705-5705
- PubMed: 33177498 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-020-19456-8
- Primary Citation Related Structures:
7ANZ - PubMed Abstract:
The nucleation of microtubules from αβ-tubulin subunits is mediated by γ-tubulin complexes, which vary in composition across organisms. Aiming to understand how de novo microtubule formation is achieved and regulated by a minimal microtubule nucleation system, we here determined the cryo-electron microscopy structure of the heterotetrameric γ-tubulin small complex (γ-TuSC) from C. albicans at near-atomic resolution. Compared to the vertebrate γ-tubulin ring complex (γ-TuRC), we observed a vastly remodeled interface between the SPC/GCP-γ-tubulin spokes, which stabilizes the complex and defines the γ-tubulin arrangement. The relative positioning of γ-tubulin subunits indicates that a conformational rearrangement of the complex is required for microtubule nucleation activity, which follows opposing directionality as predicted for the vertebrate γ-TuRC. Collectively, our data suggest that the assembly and regulation mechanisms of γ-tubulin complexes fundamentally differ between the microtubule nucleation systems in lower and higher eukaryotes.
- Zentrum für Molekulare Biologie der Universität Heidelberg, DKFZ-ZMBH Allianz, Im Neuenheimer Feld 282, 69120, Heidelberg, Germany.
Organizational Affiliation: