9.8 MAG. A new host lipid for in meso (lipid cubic phase) crystallization of integral membrane proteins
van Dalsen, L., Smithers, L., Boland, C., Weichert, D., Caffrey, M.(2020) Cryst Growth Des
Experimental Data Snapshot
Starting Model: experimental
View more details
(2020) Cryst Growth Des
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Apolipoprotein N-acyltransferase | 532 | Escherichia coli | Mutation(s): 0 Gene Names: lnt, ACU57_00505, AM464_20560, AUQ13_21565, BMA87_17500, BUE81_17200, BvCms2454_02009, BvCmsHHP001_00880, BvCmsKSNP120_02778, BvCmsKSP076_04015... EC: 2.3.1.269 Membrane Entity: Yes | 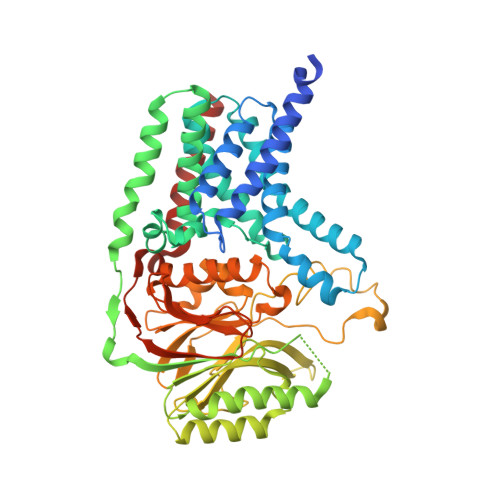 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P23930 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| LH9 (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | H [auth A] I [auth A] J [auth A] K [auth A] L [auth A] | [(2~{S})-2,3-bis(oxidanyl)propyl] heptadec-9-enoate C20 H38 O4 NXAQGVNJNZDCNZ-IBGZPJMESA-N |  | ||
| GOL Download:Ideal Coordinates CCD File | B [auth A] C [auth A] D [auth A] E [auth A] F [auth A] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 48.775 | α = 90 |
| b = 76.173 | β = 90 |
| c = 156.531 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| Coot | model building |
| XDS | data reduction |
| Aimless | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Science Foundation Ireland | Ireland | -- |