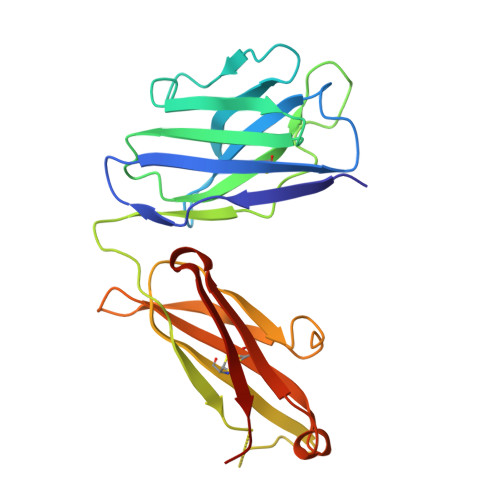
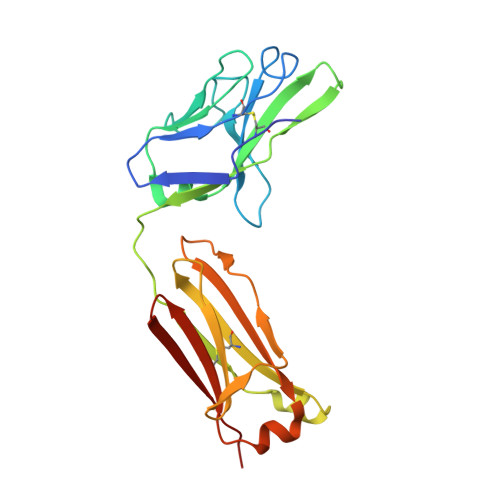
Multimeric antibodies from antigen-specific human IgM+ memory B cells restrict Plasmodium parasites.
Thouvenel, C.D., Fontana, M.F., Netland, J., Krishnamurty, A.T., Takehara, K.K., Chen, Y., Singh, S., Miura, K., Keitany, G.J., Lynch, E.M., Portugal, S., Miranda, M.C., King, N.P., Kollman, J.M., Crompton, P.D., Long, C.A., Pancera, M., Rawlings, D.J., Pepper, M.(2021) J Exp Medicine 218
- PubMed: 33661302 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1084/jem.20200942
- Primary Citation Related Structures:
6XQW - PubMed Abstract:
Multimeric immunoglobulin-like molecules arose early in vertebrate evolution, yet the unique contributions of multimeric IgM antibodies to infection control are not well understood. This is partially due to the difficulty of distinguishing low-affinity IgM, secreted rapidly by plasmablasts, from high-affinity antibodies derived from later-arising memory cells. We developed a pipeline to express B cell receptors (BCRs) from Plasmodium falciparum-specific IgM+ and IgG+ human memory B cells (MBCs) as both IgM and IgG molecules. BCRs from both subsets were somatically hypermutated and exhibited comparable monomeric affinity. Crystallization of one IgM+ MBC-derived antibody complexed with antigen defined a linear epitope within a conserved Plasmodium protein. In its physiological multimeric state, this antibody displayed exponentially higher antigen binding than a clonally identical IgG monomer, and more effectively inhibited P. falciparum invasion. Forced multimerization of this IgG significantly improved both antigen binding and parasite restriction, underscoring how avidity can alter antibody function. This work demonstrates the potential of high-avidity IgM in both therapeutics and vaccines.
- Center for Immunity and Immunotherapies, Seattle Children's Research Institute, Seattle, WA.
Organizational Affiliation: