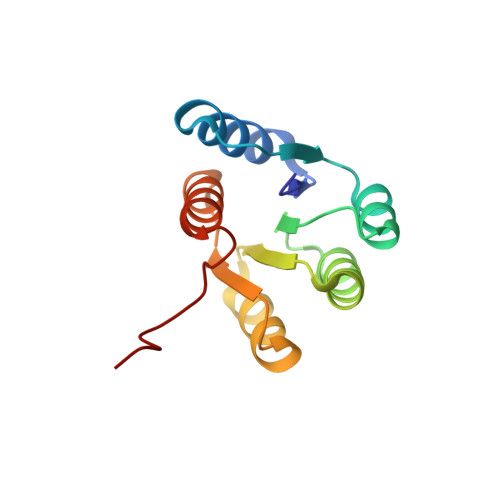
Role of backbone strain in de novo design of complex alpha/beta protein structures Accurate de novo design of asymetric alpha/beta proteins with ten or more secondary structure elements requires consideration of backbone strain Design principle proposed from designed larger alpha/beta-proteins not folded as designed: Consistency between local, non-local, and global structures
Koga, N., Koga, R., Liu, G., Castellanos, J., Montelione, G.T., Baker, D.To be published.