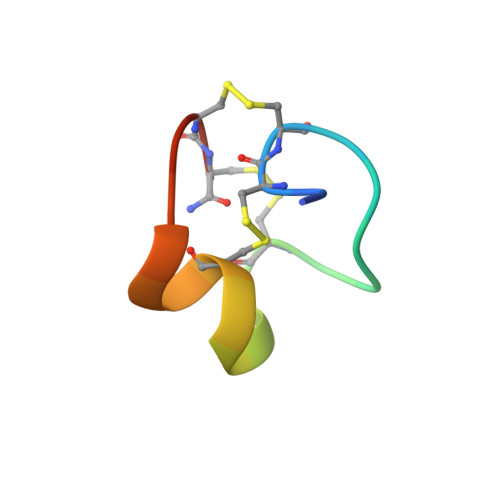
Discovery, Pharmacological Characterisation and NMR Structure of the Novel μ-Conotoxin SxIIIC, a Potent and Irreversible Na V Channel Inhibitor.
McMahon, K.L., Tran, H.N.T., Deuis, J.R., Lewis, R.J., Vetter, I., Schroeder, C.I.(2020) Biomedicines 8
- PubMed: 33023152 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.3390/biomedicines8100391
- Primary Citation Related Structures:
6X8R - PubMed Abstract:
Voltage-gated sodium (Na V ) channel subtypes, including Na V 1.7, are promising targets for the treatment of neurological diseases, such as chronic pain. Cone snail-derived µ-conotoxins are small, potent Na V channel inhibitors which represent potential drug leads. Of the 22 µ-conotoxins characterised so far, only a small number, including KIIIA and CnIIIC, have shown inhibition against human Na V 1.7. We have recently identified a novel µ-conotoxin, SxIIIC, from Conus striolatus . Here we present the isolation of native peptide, chemical synthesis, characterisation of human Na V channel activity by whole-cell patch-clamp electrophysiology and analysis of the NMR solution structure. SxIIIC displays a unique Na V channel selectivity profile (1.4 > 1.3 > 1.1 ≈ 1.6 ≈ 1.7 > 1.2 >> 1.5 ≈ 1.8) when compared to other µ-conotoxins and represents one of the most potent human Na V 1.7 putative pore blockers (IC 50 152.2 ± 21.8 nM) to date. NMR analysis reveals the structure of SxIIIC includes the characteristic α-helix seen in other µ-conotoxins. Future investigations into structure-activity relationships of SxIIIC are expected to provide insights into residues important for Na V channel pore blocker selectivity and subsequently important for chronic pain drug development.
- Institute for Molecular Bioscience, The University of Queensland, Brisbane, QLD 4072, Australia.
Organizational Affiliation: