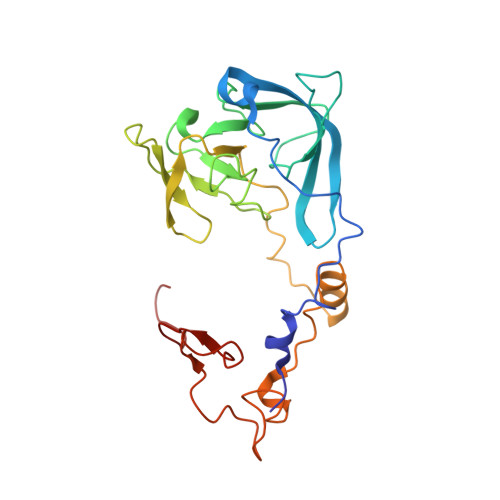
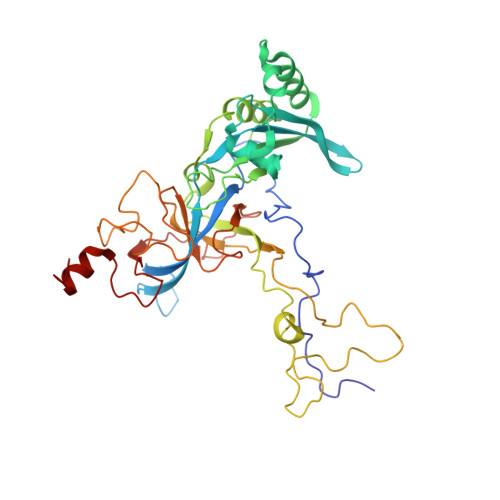
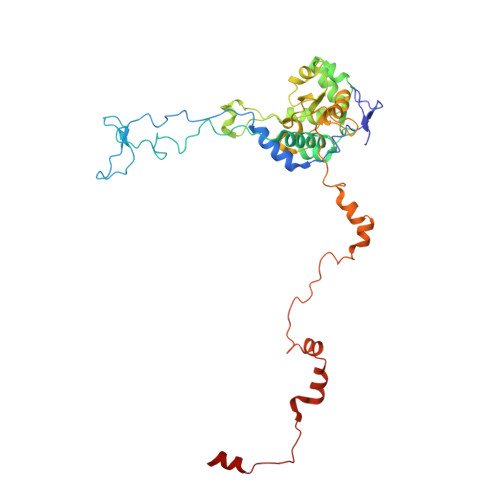
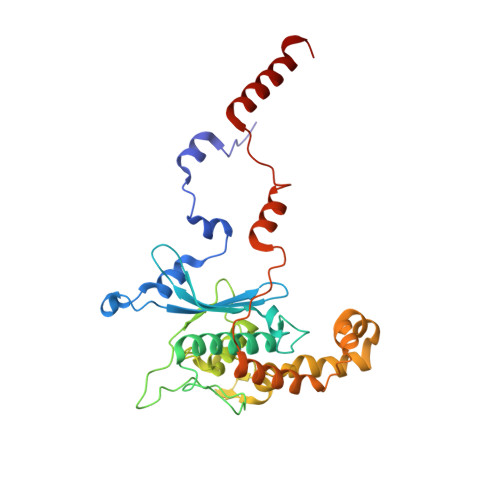
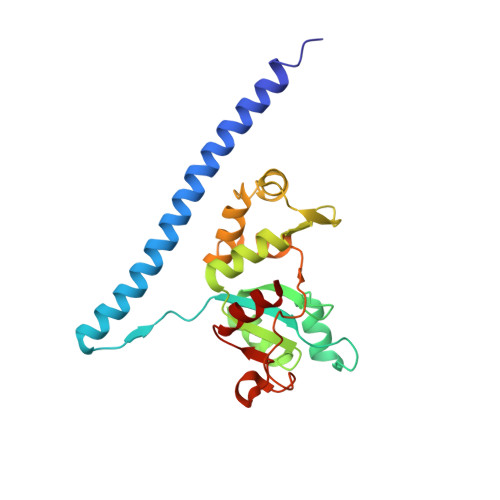
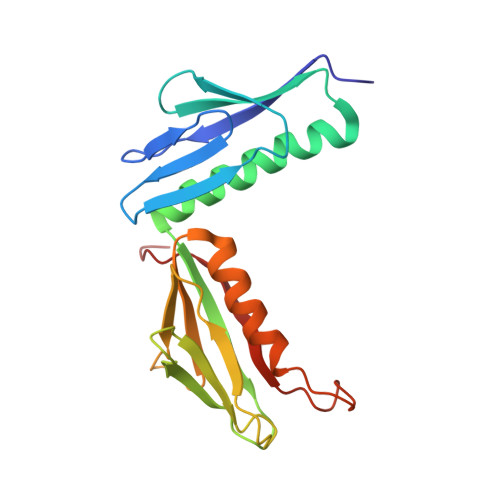
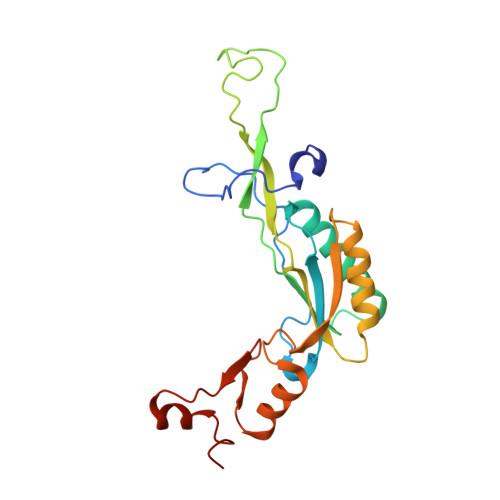
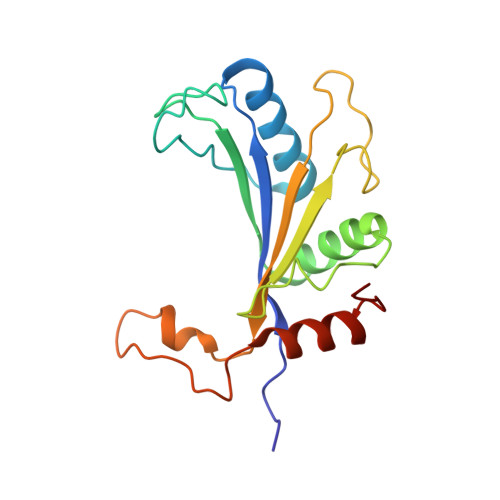
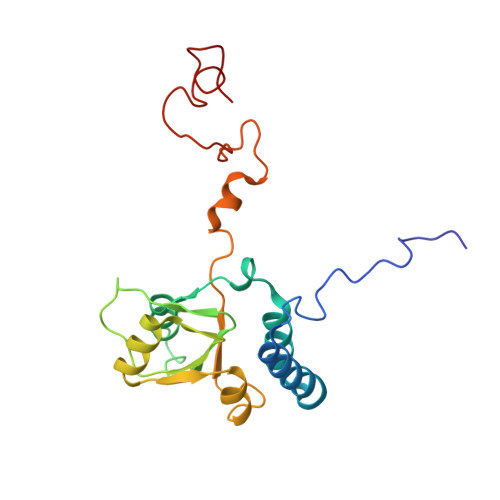
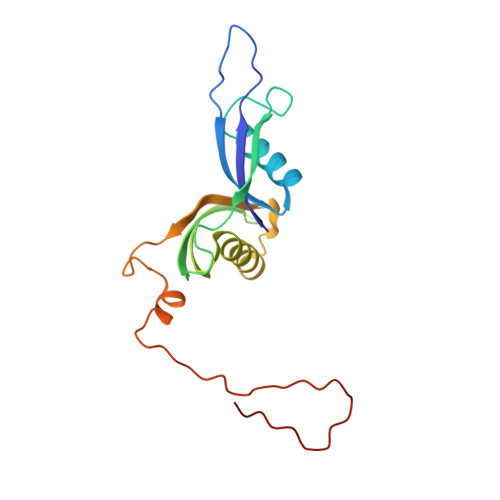
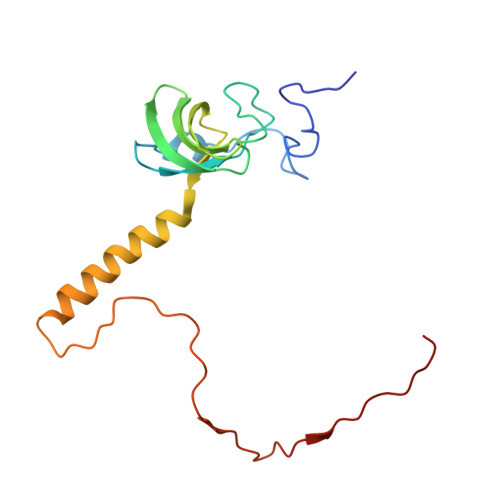
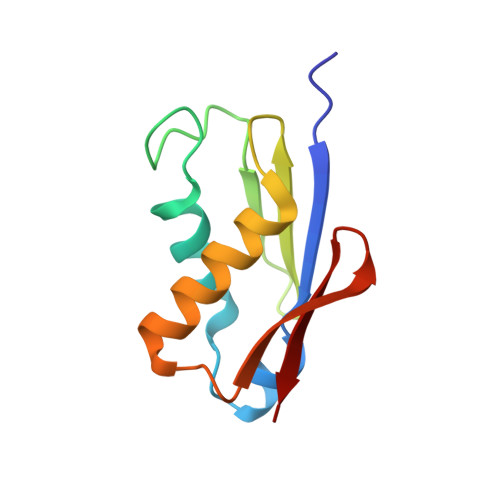
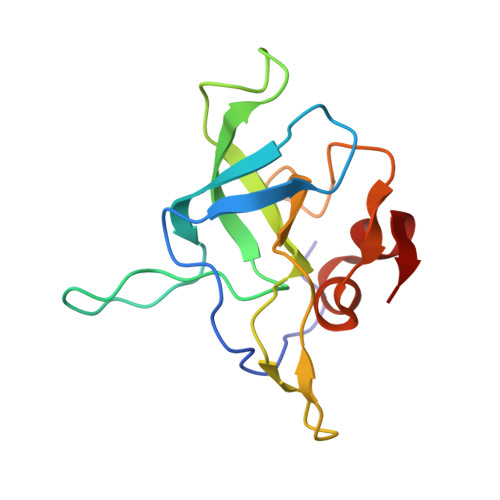
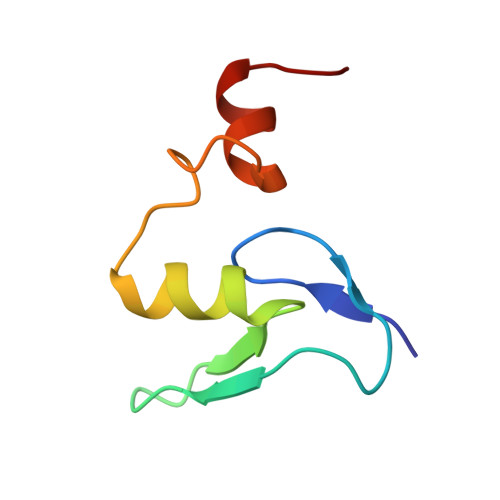
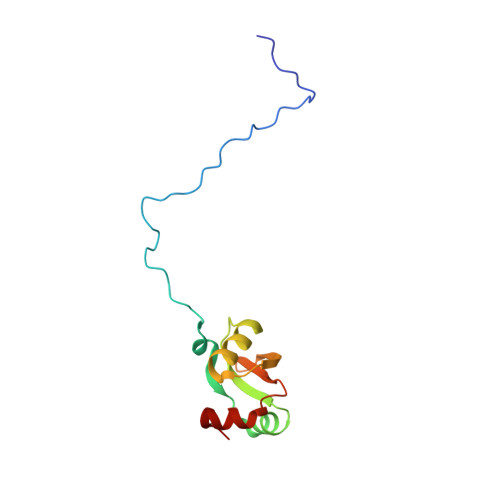
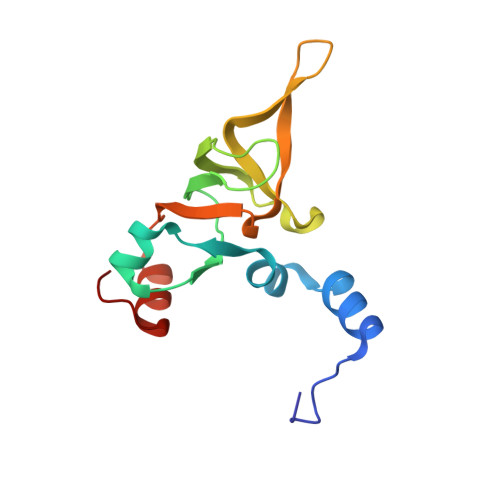
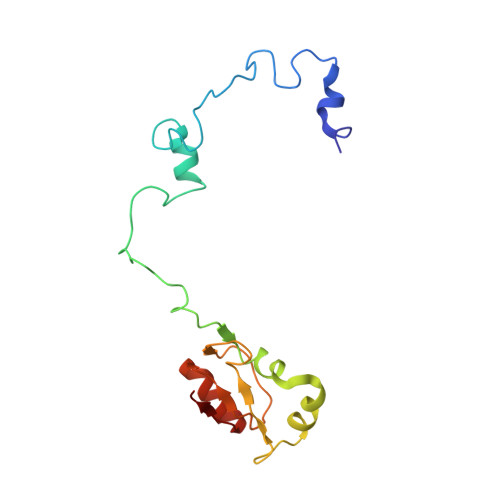
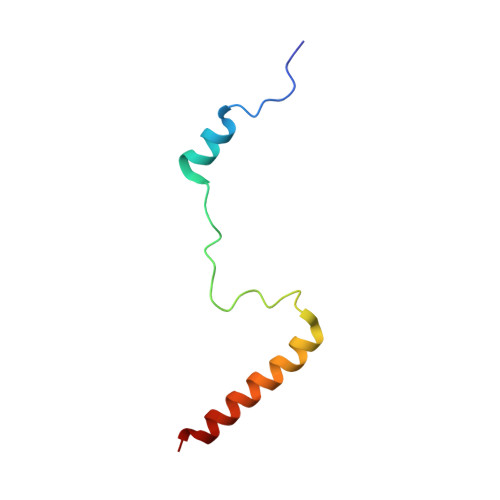
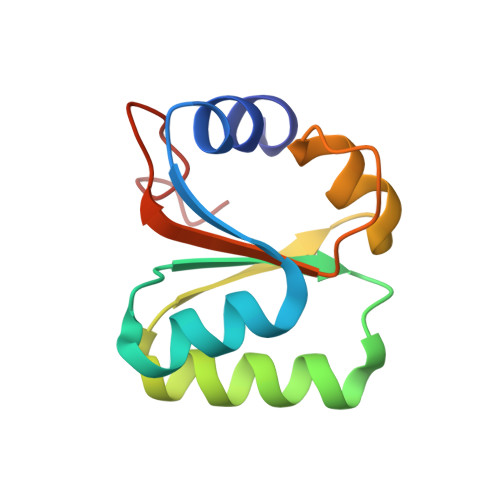
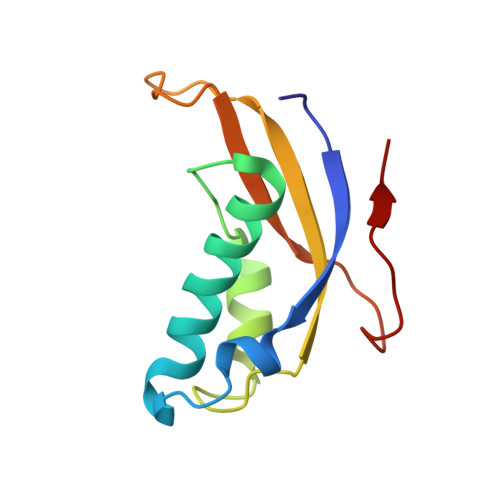
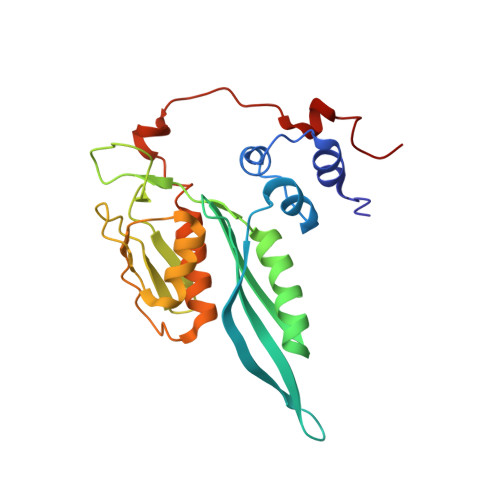
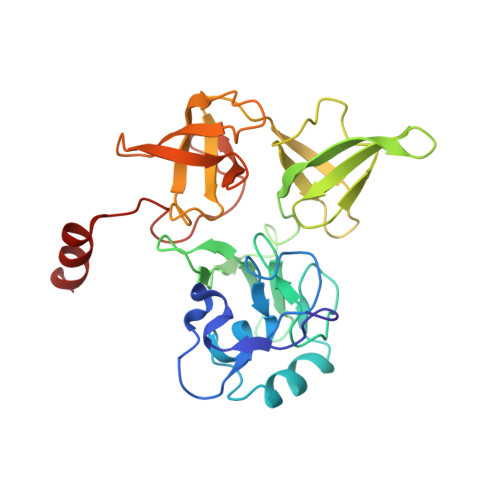
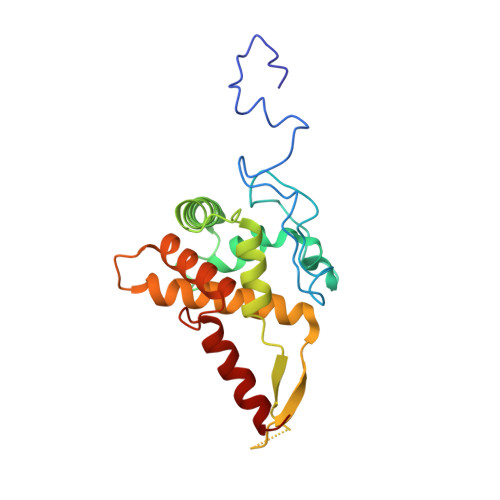
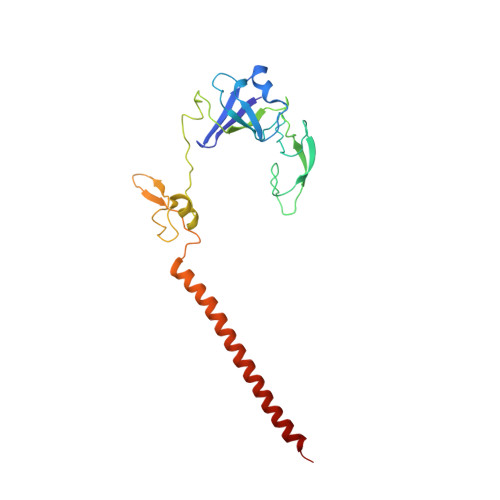
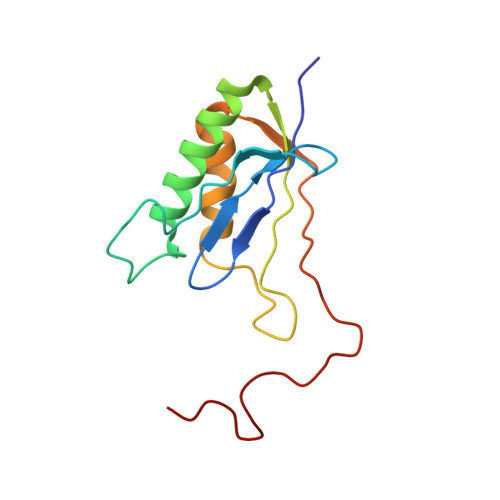
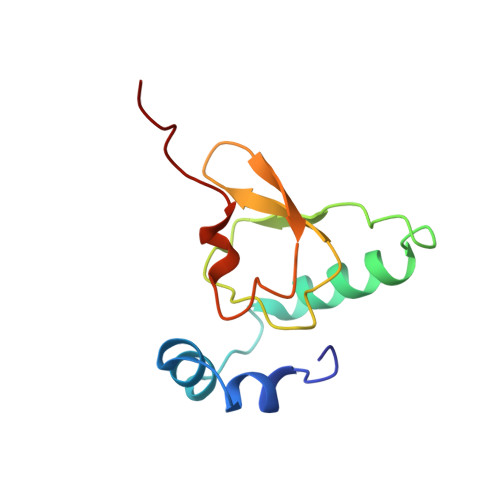
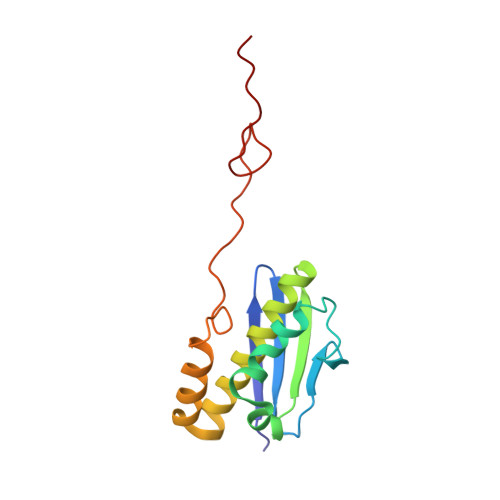
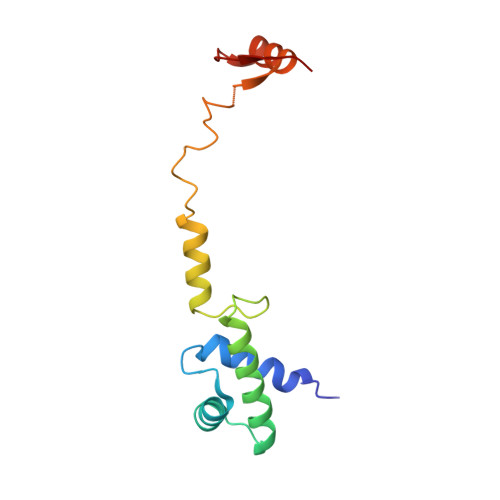
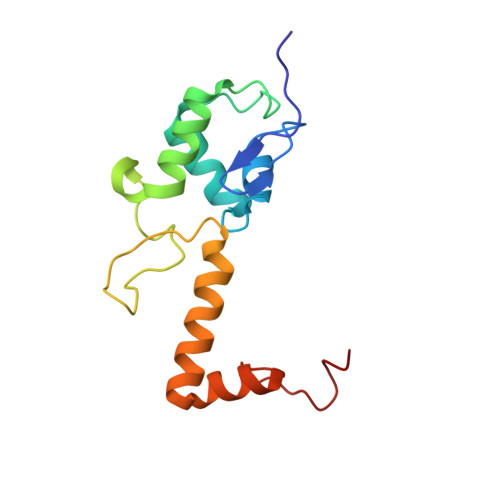
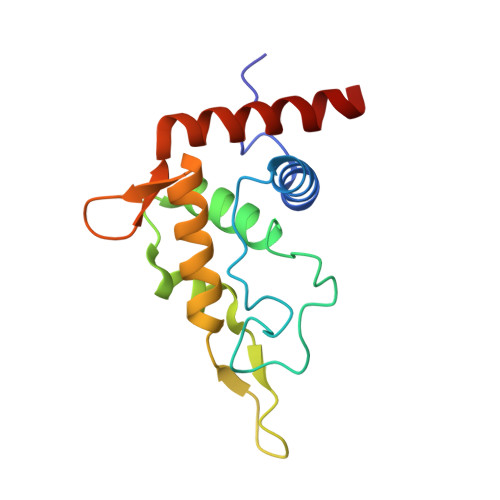
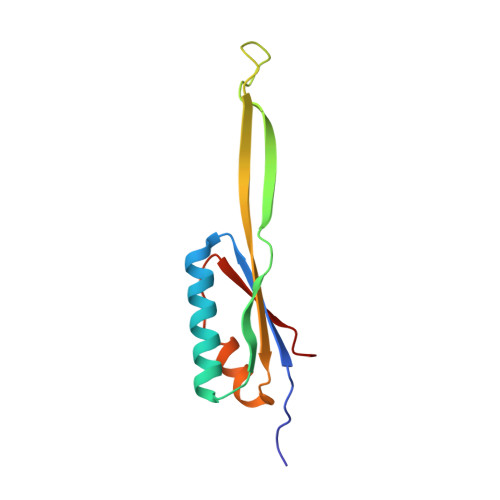
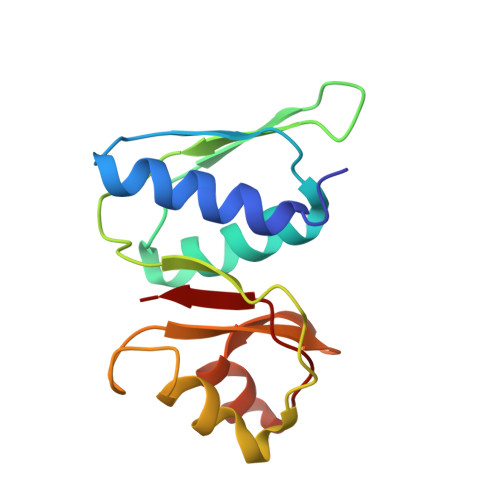
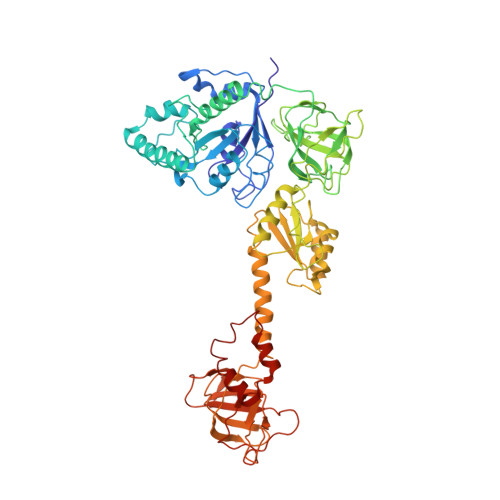
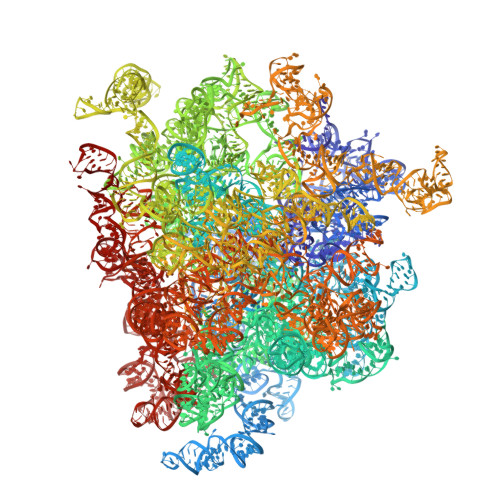
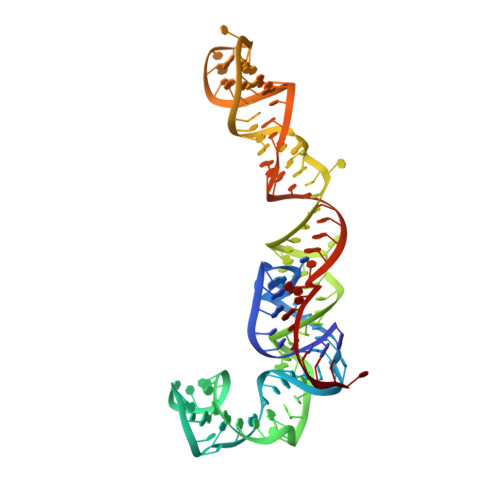
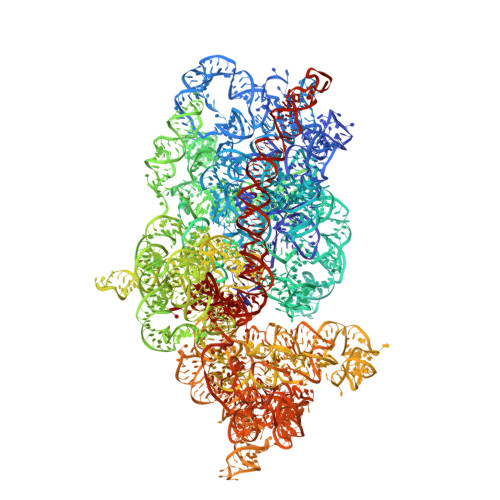
Structural basis for the transition from translation initiation to elongation by an 80S-eIF5B complex.
Wang, J., Wang, J., Shin, B.S., Kim, J.R., Dever, T.E., Puglisi, J.D., Fernandez, I.S.(2020) Nat Commun 11: 5003-5003
- PubMed: 33024099 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-020-18829-3
- Primary Citation Related Structures:
6WOO - PubMed Abstract:
Recognition of a start codon by the initiator aminoacyl-tRNA determines the reading frame of messenger RNA (mRNA) translation by the ribosome. In eukaryotes, the GTPase eIF5B collaborates in the correct positioning of the initiator Met-tRNA i Met on the ribosome in the later stages of translation initiation, gating entrance into elongation. Leveraging the long residence time of eIF5B on the ribosome recently identified by single-molecule fluorescence measurements, we determine the cryoEM structure of the naturally long-lived ribosome complex with eIF5B and Met-tRNA i Met immediately before transition into elongation. The structure uncovers an unexpected, eukaryotic specific and dynamic fidelity checkpoint implemented by eIF5B in concert with components of the large ribosomal subunit.
- Department of Structural Biology, Stanford University School of Medicine, Stanford, CA, USA.
Organizational Affiliation: