Crystal structure of prephenate dehydratase from Brucella melitensis biovar abortus 2308 in complex with phenylalanine
Abendroth, J., Dranow, D.M., Lorimer, D.D., Bullen, J.C., Sankaran, B., Horanyi, P.S., Edwards, T.E.To be published.
Experimental Data Snapshot
Starting Models: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Prephenate dehydratase:Amino acid-binding ACT | 308 | Brucella abortus 2308 | Mutation(s): 0 Gene Names: pheA, BAB1_0034 EC: 4.2.1.51 | 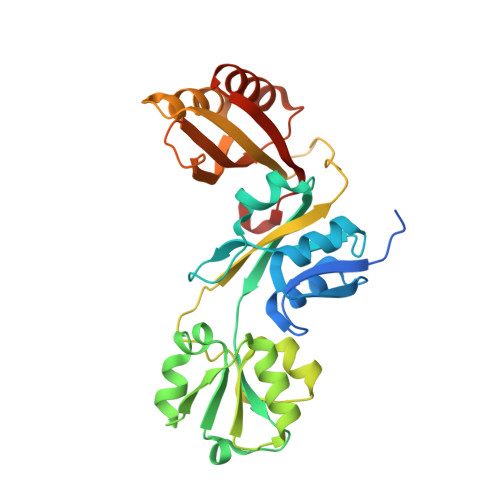 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q2YPM5 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| PHE Download:Ideal Coordinates CCD File | E [auth A], G [auth B], J [auth C], L [auth D] | PHENYLALANINE C9 H11 N O2 COLNVLDHVKWLRT-QMMMGPOBSA-N |  | ||
| EDO Download:Ideal Coordinates CCD File | F [auth A] H [auth B] I [auth B] K [auth C] M [auth D] | 1,2-ETHANEDIOL C2 H6 O2 LYCAIKOWRPUZTN-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 226.95 | α = 90 |
| b = 61.25 | β = 112.95 |
| c = 120.05 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| XDS | data reduction |
| XSCALE | data scaling |
| PDB_EXTRACT | data extraction |
| MR-Rosetta | phasing |