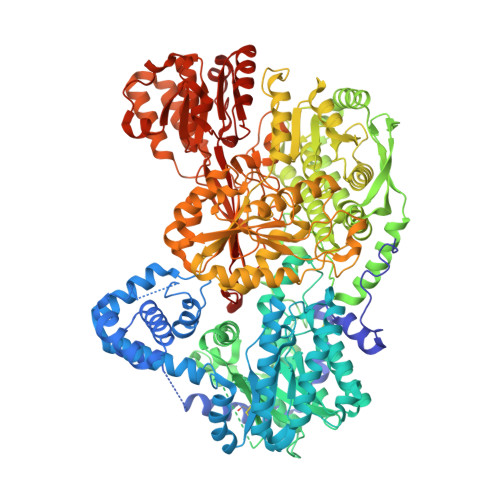
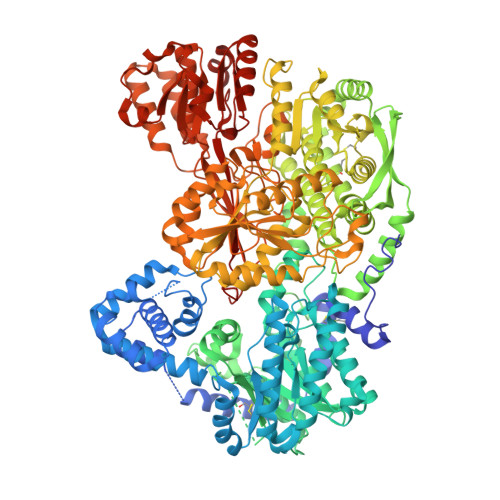
Covalent Modification of the Flavin in Proline Dehydrogenase by Thiazolidine-2-Carboxylate.
Campbell, A.C., Becker, D.F., Gates, K.S., Tanner, J.J.(2020) ACS Chem Biol 15: 936-944
- PubMed: 32159324 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1021/acschembio.9b00935
- Primary Citation Related Structures:
6UFP, 6VZ9 - PubMed Abstract:
Proline dehydrogenase (PRODH) catalyzes the first step of proline catabolism, the FAD-dependent 2-electron oxidation of l-proline to Δ 1 -pyrroline-5-carboxylate. PRODH has emerged as a possible cancer therapy target, and thus the inhibition of PRODH is of interest. Here we show that the proline analogue thiazolidine-2-carboxylate (T2C) is a mechanism-based inactivator of PRODH. Structures of the bifunctional proline catabolic enzyme proline utilization A (PutA) determined from crystals grown in the presence of T2C feature strong electron density for a 5-membered ring species resembling l-T2C covalently bound to the N5 of the FAD in the PRODH domain. The modified FAD exhibits a large butterfly bend angle, indicating that the FAD is locked into the 2-electron reduced state. Reduction of the FAD is consistent with the crystals lacking the distinctive yellow color of the oxidized enzyme and stopped-flow kinetic data showing that T2C is a substrate for the PRODH domain of PutA. A mechanism is proposed in which PRODH catalyzes the oxidation of T2C at the C atom adjacent to the S atom of the thiazolidine ring (C5). Then, the N5 atom of the reduced FAD attacks the C5 of the oxidized T2C species, resulting in the covalent adduct observed in the crystal structure. To our knowledge, this is the first report of T2C inactivating (or inhibiting) PRODH or any other flavoenzyme. These results may inform the design of new mechanism-based inactivators of PRODH for use as chemical probes to study the roles of proline metabolism in cancer.
- Department of Biochemistry, University of Missouri, Columbia, Missouri 65211, United States.
Organizational Affiliation: