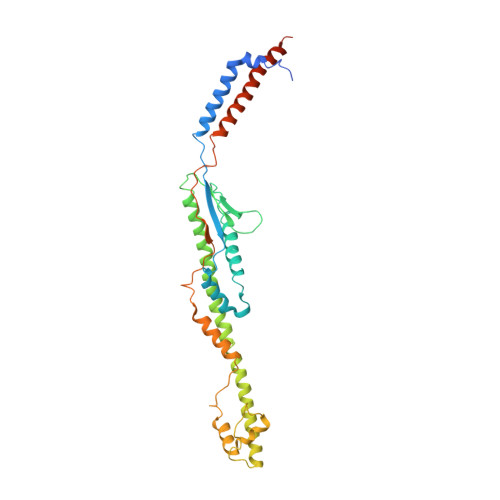
Structure of a full-length bacterial polysaccharide co-polymerase.
Wiseman, B., Nitharwal, R.G., Widmalm, G., Hogbom, M.(2021) Nat Commun 12: 369-369
- PubMed: 33446644 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-020-20579-1
- Primary Citation Related Structures:
6RBG - PubMed Abstract:
Lipopolysaccharides are important components of the bacterial cell envelope that among other things act as a protective barrier against the environment and toxic molecules such as antibiotics. One of the most widely disseminated pathways of polysaccharide biosynthesis is the inner membrane bound Wzy-dependent pathway. Here we present the 3.0 Å structure of the co-polymerase component of this pathway, WzzB from E. coli solved by single-particle cryo-electron microscopy. The overall architecture is octameric and resembles a box jellyfish containing a large bell-shaped periplasmic domain with the 2-helix transmembrane domain from each protomer, positioned 32 Å apart, encircling a large empty transmembrane chamber. This structure also reveals the architecture of the transmembrane domain, including the location of key residues for the Wzz-family of proteins and the Wzy-dependent pathway present in many Gram-negative bacteria, explaining several of the previous biochemical and mutational studies and lays the foundation for future investigations.
- Department of Biochemistry and Biophysics, Stockholm University, Stockholm, Sweden. bwise@dbb.su.se.
Organizational Affiliation: