Ultracompact 3D microfluidics for time-resolved structural biology.
Knoska, J., Adriano, L., Awel, S., Beyerlein, K.R., Yefanov, O., Oberthuer, D., Pena Murillo, G.E., Roth, N., Sarrou, I., Villanueva-Perez, P., Wiedorn, M.O., Wilde, F., Bajt, S., Chapman, H.N., Heymann, M.(2020) Nat Commun 11: 657-657
- PubMed: 32005876 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-020-14434-6
- Primary Citation Related Structures:
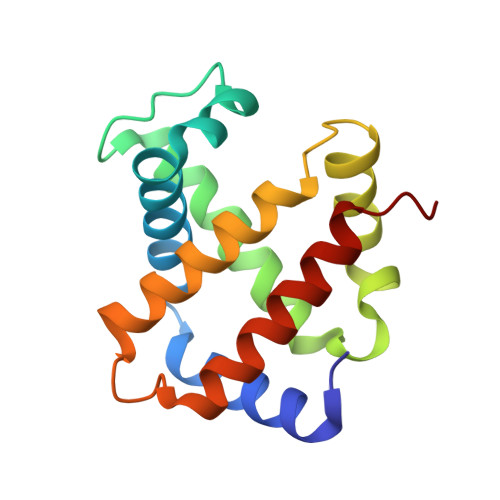
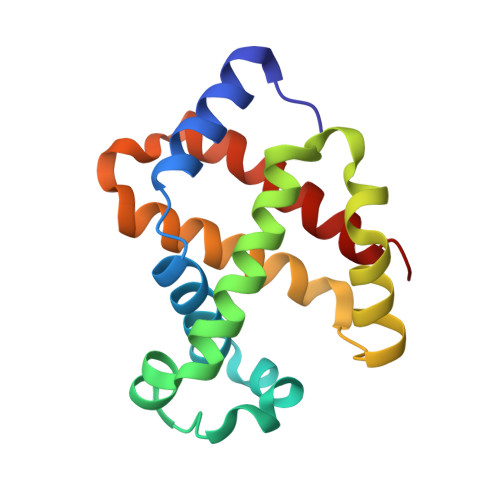
6R2O - PubMed Abstract:
To advance microfluidic integration, we present the use of two-photon additive manufacturing to fold 2D channel layouts into compact free-form 3D fluidic circuits with nanometer precision. We demonstrate this technique by tailoring microfluidic nozzles and mixers for time-resolved structural biology at X-ray free-electron lasers (XFELs). We achieve submicron jets with speeds exceeding 160 m s -1 , which allows for the use of megahertz XFEL repetition rates. By integrating an additional orifice, we implement a low consumption flow-focusing nozzle, which is validated by solving a hemoglobin structure. Also, aberration-free in operando X-ray microtomography is introduced to study efficient equivolumetric millisecond mixing in channels with 3D features integrated into the nozzle. Such devices can be printed in minutes by locally adjusting print resolution during fabrication. This technology has the potential to permit ultracompact devices and performance improvements through 3D flow optimization in all fields of microfluidic engineering.
- CFEL, Center for Free-Electron Laser Science, Deutsches Elektronen-Synchrotron DESY, Notkestrasse 85, 22607, Hamburg, Germany.
Organizational Affiliation: