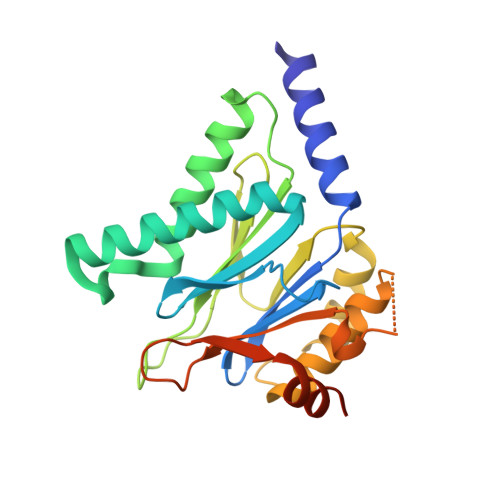
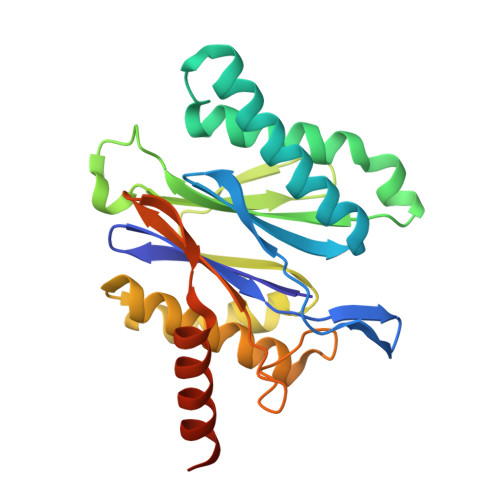
Selective Phenylimidazole-Based Inhibitors of theMycobacterium tuberculosisProteasome.
Zhan, W., Hsu, H.C., Morgan, T., Ouellette, T., Burns-Huang, K., Hara, R., Wright, A.G., Imaeda, T., Okamoto, R., Sato, K., Michino, M., Ramjee, M., Aso, K., Meinke, P.T., Foley, M., Nathan, C.F., Li, H., Lin, G.(2019) J Med Chem 62: 9246-9253
- PubMed: 31560200 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1021/acs.jmedchem.9b01187
- Primary Citation Related Structures:
6OCW, 6OCZ, 6ODE - PubMed Abstract:
Proteasomes of pathogenic microbes have become attractive targets for anti-infectives. Coevolving with its human host, Mycobacterium tuberculosis (Mtb) has developed mechanisms to resist host-imposed nitrosative and oxidative stresses. Genetic deletion or pharmacological inhibition of the Mtb proteasome (Mtb20S) renders nonreplicating Mtb susceptible to reactive nitrogen species in vitro and unable to survive in the lungs of mice, validating the Mtb proteasome as a promising target for anti-Mtb agents. Using a structure-guided and flow chemistry-enabled study of structure-activity relationships, we developed phenylimidazole-based peptidomimetics that are highly potent for Mtb20S. X-ray structures of selected compounds with Mtb20S shed light on their selectivity for mycobacterial over human proteasomes.
- Department of Microbiology & Immunology , Weill Cornell Medicine , 1300 York Avenue , New York , New York 10065 , United States.
Organizational Affiliation: