Crystal Structure of Selenocysteine Lyase from Escherichia coli
Scortecci, J.F., Brandao-Neto, J., Pereira, H.M., Thiemann, O.H.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cysteine desulfurase | 406 | Escherichia coli UMEA 3718-1 | Mutation(s): 0 Gene Names: sufS, G994_01747 EC: 2.8.1.7 (PDB Primary Data), 4.4.1.16 (PDB Primary Data) | 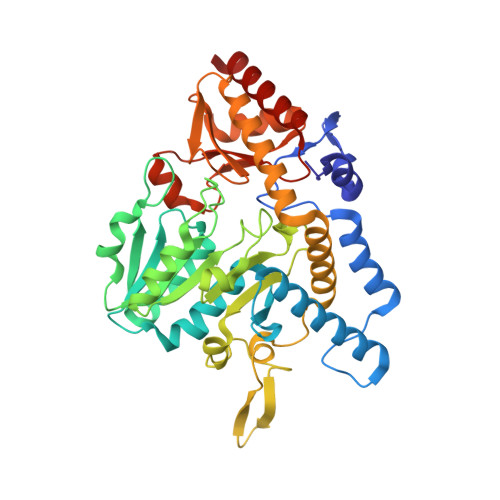 | |
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| PLP (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | B [auth A] | PYRIDOXAL-5'-PHOSPHATE C8 H10 N O6 P NGVDGCNFYWLIFO-UHFFFAOYSA-N |  | ||
| NA Download:Ideal Coordinates CCD File | C [auth A] | SODIUM ION Na FKNQFGJONOIPTF-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 126.52 | α = 90 |
| b = 126.52 | β = 90 |
| c = 134.091 | γ = 90 |
| Software Name | Purpose |
|---|---|
| xia2 | data scaling |
| PHASER | phasing |
| PHENIX | refinement |
| PDB_EXTRACT | data extraction |
| xia2 | data reduction |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Sao Paulo Research Foundation (FAPESP) | Brazil | 2016/20977-7 |