Calcium-Binding Generates the Semi-Clathrate Waters on a Type II Antifreeze Protein to Adsorb onto an Ice Crystal Surface.
Arai, T., Nishimiya, Y., Ohyama, Y., Kondo, H., Tsuda, S.(2019) Biomolecules 9
- PubMed: 31035615 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.3390/biom9050162
- Primary Citation Related Structures:
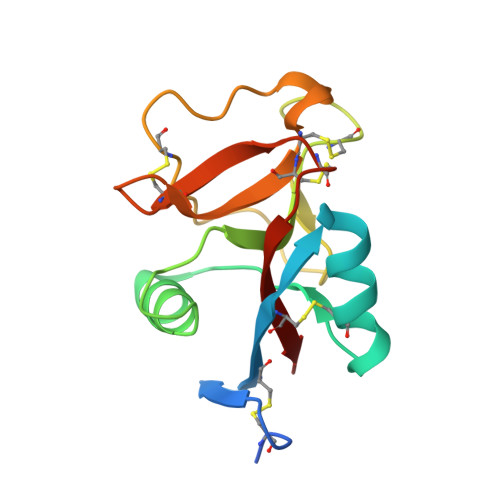
6JK4, 6JK5 - PubMed Abstract:
Hydration is crucial for a function and a ligand recognition of a protein. The hydration shell constructed on an antifreeze protein (AFP) contains many organized waters, through which AFP is thought to bind to specific ice crystal planes. For a Ca 2+ -dependent species of AFP, however, it has not been clarified how 1 mol of Ca 2+ -binding is related with the hydration and the ice-binding ability. Here we determined the X-ray crystal structure of a Ca 2+ -dependent AFP (jsAFP) from Japanese smelt, Hypomesus nipponensis , in both Ca 2+ -bound and -free states. Their overall structures were closely similar (Root mean square deviation (RMSD) of Cα = 0.31 Å), while they exhibited a significant difference around their Ca 2+ -binding site. Firstly, the side-chains of four of the five Ca 2+ -binding residues (Q92, D94 E99, D113, and D114) were oriented to be suitable for ice binding only in the Ca 2+ -bound state. Second, a Ca 2+ -binding loop consisting of a segment D94-E99 becomes less flexible by the Ca 2+ -binding. Third, the Ca 2+ -binding induces a generation of ice-like clathrate waters around the Ca 2+ -binding site, which show a perfect position-match to the waters constructing the first prism plane of a single ice crystal. These results suggest that generation of ice-like clathrate waters induced by Ca 2+ -binding enables the ice-binding of this protein.
- Graduate School of Life Science, Hokkaido University, Sapporo 060-0810, Japan. tatarai0926@gmail.com.
Organizational Affiliation: