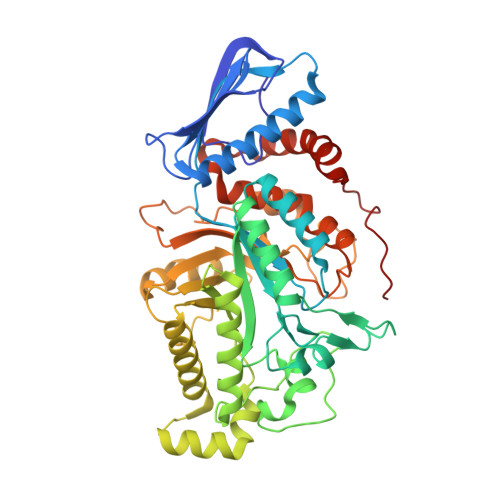
Crystallographic evidence for substrate-assisted catalysis of beta-N-acetylhexosaminidas from Akkermansia muciniphila.
Chen, X., Wang, J., Liu, M., Yang, W., Wang, Y., Tang, R., Zhang, M.(2019) Biochem Biophys Res Commun 511: 833-839
- PubMed: 30846208 Search on PubMed
- DOI: https://doi.org/10.1016/j.bbrc.2019.02.074
- Primary Citation Related Structures:
6JE8, 6JEA, 6JEB - PubMed Abstract:
β-N-acetylhexosaminidases from Akkermansia muciniphila was reported to perform the crystal structure with GlcNAc complex, which proved to be the substrate of Am2301. Domain II of Am2301 is consisted of amino acid residues 111-489 and is folded as a (β/α) 8 barrel with the active site combined of the glycosyl hydrolases. Crystallographic evidence showed that Asp-278 and Glu-279 could be the catalytic site and Tyr-373 may plays a role on binding the substrate. Moreover, Am2301 prefers to bind Zn ion, which similar to other GH 20 family. Enzyme activity and kinetic parameters of wild-type Am2301 and mutants proved that Asp-278 and Glu-279 are the catalytic sites and they play a critical role on the catalytic process. Overall, our results demonstrate that Am2301 and its complex with GlcNAC provide obvious structural evidence for substrate-assisted catalysis. Obviously, this expands our understanding on the mode of substrate-assisted reaction for this enzyme family in Akkermansia muciniphila.
- School of Life Sciences, Anhui University, 111 Jiulong Road, Hefei, Anhui, 230601, China; Key Laboratory of Human Microenvironment and Precision Medicine of Anhui Higher Education Institutes, Anhui University, 111 Jiulong Road, Hefei, Anhui, 230601, China; Department of Biological and Food Engineering, Bozhou University, 2266 Tangwang Road, Bozhou, Anhui, China.
Organizational Affiliation: