Complex structure of the HRP3 PWWP domain with both a 16-bp TA-rich DNA and a H3K36me2-containing histone peptide
Wang, Z., Tian, W.(2019) Nucleic Acids Res
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Hepatoma-derived growth factor-related protein 3 | 100 | Homo sapiens | Mutation(s): 0 Gene Names: HDGFL3, HDGF2, HDGFRP3, CGI-142 | 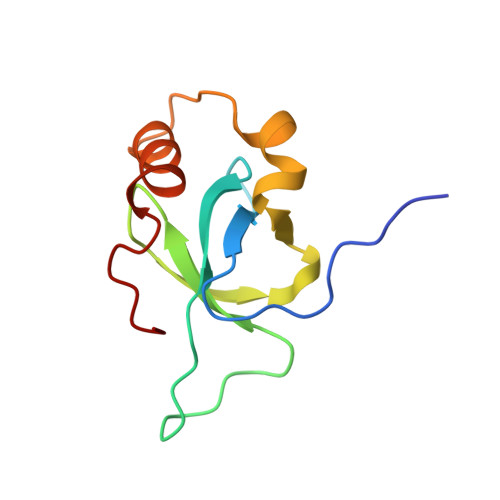 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q9Y3E1 GTEx: ENSG00000166503 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9Y3E1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| H3K36me2-containing histone peptide | 8 | synthetic construct | Mutation(s): 0 | 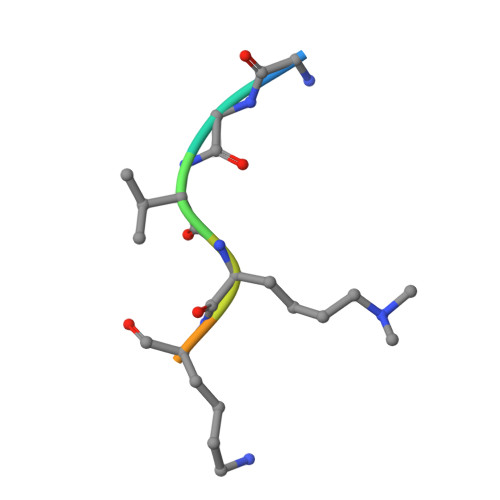 | |
UniProt & NIH Common Fund Data Resources | |||||
Entity Groups | |||||
| UniProt Group | P68431 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | ||||
| Molecule | Chains | Length | Organism | Image |
|---|---|---|---|---|
| 16-bp TA-rich DNA | 16 | synthetic construct | 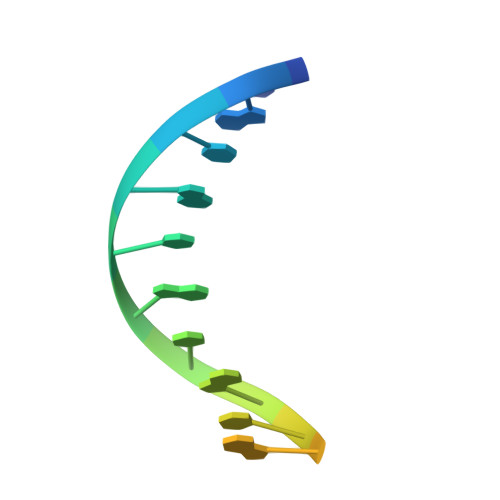 | |
Sequence AnnotationsExpand | ||||
Reference Sequence | ||||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| MLY Query on MLY | E, F | L-PEPTIDE LINKING | C8 H18 N2 O2 |  | LYS |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 33.592 | α = 90 |
| b = 33.592 | β = 90 |
| c = 200.101 | γ = 120 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |
| PHENIX | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Natural Science Foundation of China | China | 31570729 |